Translate this page into:
Oral prostacycline analog and clopidogrel combination provides early maturation and long-term survival after arteriovenous fistula creation: A randomized controlled study
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Vascular access is used as a lifeline for hemodialysis in patients with end stage renal disease failure (ESRD). Failure of arteriovenous fistula (AVF) maturation is still high. The purpose of this study was to research the effects of clopidogrel in combination with oral iloprost, a synthetic analog of prostacyclin PGI2. Ninety-six diabetic ESRD patients were divided into two groups. In the first group (Group 1, N = 50), clopidogrel (75 mg daily dose) and an oral prostacycline analog (200 mg daily dose) were administered. In the second group (Group 2, N = 46), placebo was given. All patients took study medication 7–10 days prior to surgery. A Doppler ultrasound (USG) was performed for measurement of arterial and venous diameters, and peak systolic velocity of arterial flow based on subsequent fistula adequacy. Autogenous AVFs were constructed in forearm as distally as possible in all patients. Both groups were followed-up for a year. In the placebo group, early AVF thrombosis was detected in two patients (4.3%). AVF maturation failure was noted in 14 patients (30.4%) in placebo group and in four patients (8%) in clopidogrel plus oral prostacycline analog group in the early postoperative period (P = 0.001). The mean maturation time was 38 ± 6.5 and 53 ± 12.8 days in study and placebo groups, respectively (P = 0.023). The mean blood flow was 352 ± 94 mL/min in placebo group and 604 ± 125 mL/min in study group (P = 0.001). The arterial end diastolic velocity was 116 ± 14 cm/s in study group and 72 ± 21 cm/s in placebo group (P = 0.036) 1 year after the surgery. Our data indicated that clopidogrel and oral prostacycline analog combination is effective and safe for the prevention of primary AVF failure in hemodialysis patients and decreased acute and chronic thrombotic events.
Keywords
Antiplatelets
arteriovenous fistula
arteriovenous fistula maturation
prostacycline analog
vasodilation
Introduction
Arteriovenous fistula (AVF) is the preferred vascular access for hemodialysis. Early failure of fistulas due to thrombosis or inadequate maturation is a barrier to increasing the prevalence of fistulas. Despite the benefits of AVFs as a form of vascular access in end stage renal disease (ESRD) patients, considerable time is required for the fistula to mature and suitably develop into a functional format[1234] Maturation rate after autogenous AVF depends on various factors such as age, body mass index (BMI), venous and arterial diameter, venous distensibility, and arterial elasticity.[56] The main cause of AVF failure is outflow stenosis as a result of vascular intimal hyperplasia and thrombosis.[67] This formation of neointimal hyperplasia and thrombosis may be initiated by activated platelet function, endothelial cell injury and vascular smooth muscle cell proliferation, which may occur due to unphysiologic vascular anastomosis.[678] In view of these postulated mechanisms of AVF failure, possible clinical benefit of antiplatelet therapy has been proposed.[891011] Aspirin and clopidogrel have been evaluated as prophylaxis with conflicting results. Furthermore, only a few studies have addressed the safety of aspirin use after AVF creation. Van der Linden et al. demonstrated that venous potential distensibility was significantly correlated with functional maturation, especially in the group of patients with vein diameter ≤ 2 mm.[12] They suggested that vein distensibility could be measured preoperatively by plethysmography. As a result of this study, they concluded that venous line distensibility was a better predictive factor than vein size.[12]
There is no report about the effects of combination therapy after AVF creation using clopidogrel in combination with prostacycline analog as a potent vasodilator agent after autogenous AVF creation. On the basis of these considerations, we performed a study to test the hypothesis that clopidogrel as an irreversible antiplatelet and oral prostacycline analog as an arterial vasodilator and additional antiaggregant drug may increase venous distensibility and arterial elasticity and would prevent primary AVF failure in hemodialysis patients with diabetes mellitus.
Materials and Methods
We operated on 385 ESRDP for AVF creation between April 2008 and December 2013. We excluded 289 patients [Table 1], and 96 patients met the study criteria for enrollment.

To detect the quality of the brachial artery (BA) and a venous line, Doppler USG was used. Preoperative venous and arterial spectral USG showed that there were no differences when we compared arterial and venous quality and diameter.
Patients were divided into two groups. The randomization was stratified according to the medical center with a permuted block scheme, with a block size of four and an equal allocation. Eligible participants were called to the coordinating center to obtain a randomization protocol, which corresponded to a specific medication. Neither the details of the randomization sequence nor the identity of the medication assigned was known to the participant or any personnel at the participating sites. The treatment was initiated 7–10 days prior to the scheduled access surgery and continued postoperatively. After randomization, the two groups of subjects are followed in exactly the same way, and the only difference between the care with which they receive drugs, outpatient visits and follow-up calls should be those intrinsic to the treatments being compared. In the first group of patients, 7–10 days prior to the surgery, we started clopidogrel (75 mg daily) and an oral prostacycline analog (200 mg orally daily). A placebo 7–10 days prior to the surgery was started in the control group. Treatments were continued for 1 year. Participants and members of the study team were blinded to the treatment assignment. Patients in the control and study group were operated by two surgeons using the same technique and same suture materials (6/0 polypropylene suture). To provide the purse or narrowing effect of suture, the surgeons did use two 6/0 suture material. Each surgeon did equal number of AVF creation in both the groups. We noticed that the two surgeons operated on the same number of patients in both the groups.
Study drug administration began immediately after randomization. Clopidogrel and prostacycline were administered orally prior to surgery, and continued during the year. Data collection was performed at baseline, 4 weeks after fistula creation, and monthly thereafter until ascertainment of fistula suitability. Adverse events were recorded. Study medication was discontinued when there was a sign of fistula thrombosis confirmed by vascular surgeons.
In a normally developed fistula, there is a continuous thrill from the anastomosis all the way centrally along the outflow vein. The thrill gradually decreases in intensity from the anastomosis centrally. In the absence of a stenosis, we confirmed there was a soft pulse, which was easily compressible. The presence of collaterals was determined by physical examination of skin. We defined the maturation of AVF when the mean blood flow from AVF was more than 300 mL/min. and a minimum arterial end diastolic velocity was 70 cm/s by sonographic examination for both groups. Venous and arterial line diameters were calculated using sonography. If there was no sign of restriction of blood flow when the patients underwent hemodialysis, we described the hemodialysis access to be good. A failed AVF was described when we did not reach functional status. Primary patency was defined as the interval from AVF creation to abandonment due to thrombosis or low flow, inadequate for sufficient hemodialysis. Early patency or failure was defined according to a thrill and/or bruit 4 weeks after the surgery.
The antero-posterior diameter of BA was measured in the transverse dimension on gray scale sonography. Peak systolic velocity and end-diastolic velocity of the BA were measured in a representative waveform in each patient [Figure 1]. The resistive index was a measure of the downstream flow resistance and was calculated as the difference between the respective measurements obtained after fist release (post) and during fist clenching (pre). The medication information was collected at both baseline and a year later. The primary outcome measure was final AVF failure, which required the creation of a new AVF. First, we investigated the need of re-intervention after AVF due to thrombosis or occlusion during the follow-up period. Second, a bleeding event during the study period. The secondary outcome was failure of the fistula to become suitable for dialysis. Suitability was defined as use of the fistula at a dialysis machine blood pump rate of 300 mL/min. or more during 8 of 12 dialysis sessions.
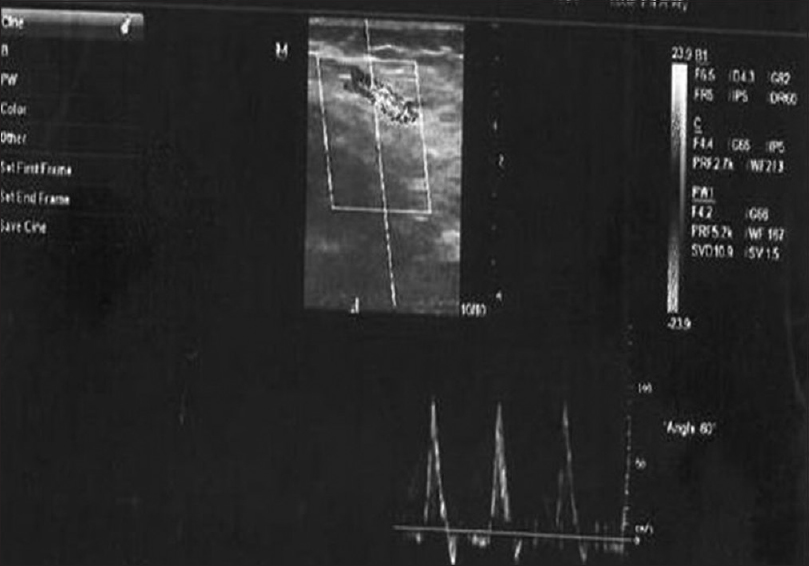
- Doppler ultrasonography (in a gray scale) shows the brachial artery and its diameter in a patient with insulin-dependent diabetes mellitus and peak velocity
Statistical analysis
The statistical program, SPSS version 13 (SPSS, Chicago, IL) was used to analyze the data. Data are presented as mean ± standard error for continuous variables and as percentages for categorical variables. All data were tested for normality using the method of Kolmogorov–Smirnov. Statistical analyses were performed on an intention-to-treat basis. The t-test was used when the means of two groups were compared. On the basis of intention-to-treat principles, all other participants were monitored according to the research protocol for whom study medications were continued. All hypothesis tests were conducted by using a significance level of 0.05 (two-sided). For evaluation of the relationships between the AVF maturation and independent variables, Chi-square test was used. Differences between matured and failed groups were tested with the Wilcoxon Rank-Sum Test for continuous variables. Kaplan-Meier survival analysis was used to calculate patency rate. Multivariate Cox proportional hazards models were used to determine factors associated with reduced AVF patency. Test results were presented as hazard ratios with 95% confidence intervals (CIs), and two-sided P < 0.05 was considered statistically significant.
Results
The mean ages were 54.23 ± 2.6 and 55.8 ± 2.8 years in Group 1 and 2 respectively (P > 0.05). There were no significant differences in covariables between groups when we compared patient characteristics, blood pressure, blood LDL, calcium, and phosphate levels [Table 2]. We did not detect any complication during the treatment period such as bleeding in Group 1 patients. Three patients complained from a temporary headache on the 1st day of oral prostacycline analog administration. However, this side effect did not require stopping of the prostacycline analog. All patients were taking study medication prior to surgery and did not have to be discontinued prematurely. None of the patients died or were lost to follow-up. Four weeks after the surgery, AVF maturation failure developed in 14 patients (30.4%) in placebo group and in 4 patients (8%) in clopidogrel and oral prostacycline analog group (P = 0.001). Fistula maturation and survival has been confirmed using physical examinations and by a Doppler USG every month for each patient. The Kaplan−Meier survival curve showed AVF maturation and survival results in Figure 2 [Table 3]. The mean maturation time of AVF was 38 ± 6.5 day and 53 ± 12.8 day in clopidogrel plus oral prostacycline analog and placebo groups, respectively (P = 0.023). Patients treated with clopidogrel and oral prostacycline analog had a increased AVF survival (P = 0.001). In the placebo group, early AVF failure was detected in two patients due to venous line thrombosis. We re-operated on these patients immediately after the detection of AVF failure.

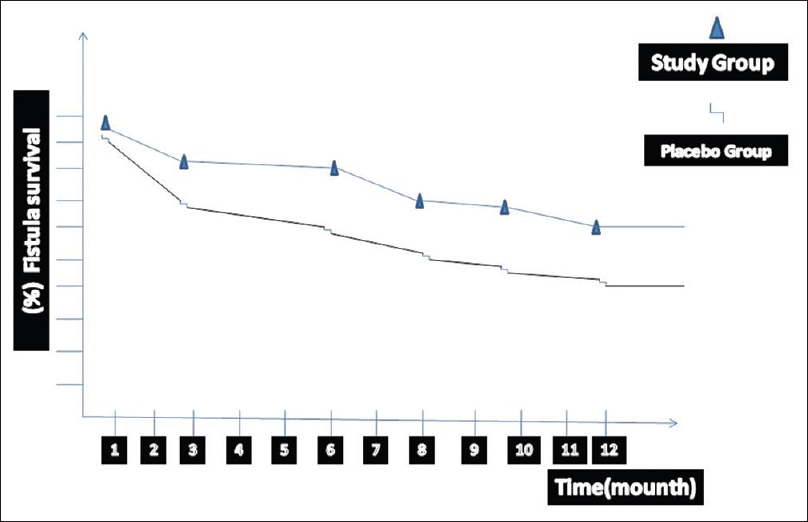
- Demonstrates the survival rate of arteriovenous fistulas (AVFs) during the study period in group 1 and group 2. Kaplan–Meier estimates of the cumulative incidence of loss of primary AVFs patency in both groups. The median duration of patency was 5.8 months (95% confidence interval (CI): 4.3–7.1) in the group 1 (study group) and 4.3 months (95% CI: 3.6–5.4) in the group 2 (placebo group)

The preoperative mean diameter of BA was 4.3 ± 1.5 mm in clopidogrel plus oral prostacycline analog group and 4.04 ± 1.3 mm in placebo group (P = 0.94). After surgery, USG showed that the mean diameter of BA was 6.3 ± 1.4 mm and 4.74 ± 0.9 mm in the study and placebo group respectively (P = 0.002). The preoperative mean diameter of the vein in the study and placebo groups were 3.1 ± 0.8 and 3.04 ± 0.7 mm (P = 0.96). In the postoperative period, the corresponding values were 5.4 ± 0.9 and 4.01 ± 0.3 mm respectively (P = 0.024). Blood flow from AVF was 352 ± 94 mL/min. in placebo group and 604 ± 125 mL/min. in clopidogrel plus oral prostacycline analog group (P = 0.001). Doppler USG examination of AVF showed that a minimum arterial end-diastolic velocity has been detected at 116 ± 14 cm/s in Group 1, 3 months after the surgery. However, this value was 72 ± 21 cm/s in the placebo group (P = 0.036).
Overall, the hazard ratio for primary AVF failure was 0.82 (95% CI: 0.31–0.94). During the follow-up period, from 4 weeks to 6 months, tenderness of the extremity, edema, and hematoma occurred in nine patients in study group, and in six in placebo group. Hemodialysis was successfully performed in 36 patients (72%) in the study group and 22 patients (47.8%) in the placebo group at the end of study (P = 0.001). The hemodialysis period in Group 1 was significantly longer and more successful than in the placebo group (P = 0.001). Multivariate logistic regression analysis revealed that AVF maturation and survival rate was more than in study group (odds ratio 2.27; 95% CI: 1.07–8.50; P = 0.001). The survival and maturation time of AVF in study group and placebo group has been summarized in Figure 3(P = 0.001). The flow diagram demonstrates the number of patients who were screened and selected for research [Figure 4]. During follow-up, we periodically confirmed the collateral vessels using physical and Doppler ultrasound examinations. There were no important symptoms related to collateral vessels that required embolization or ligation.
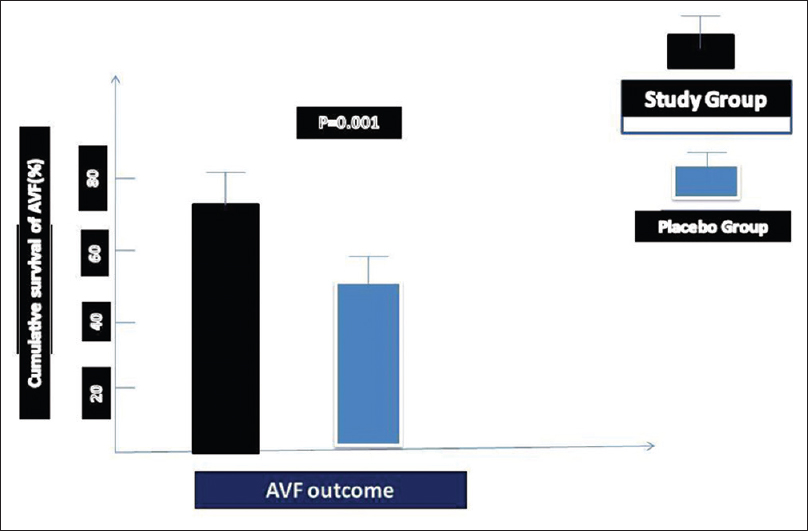
- Cumulative incidence for primary end points. This graphic demonstrates the cumulative incidence for the percent of primary end points attributable to flow monitoring compared to total end points in each treatment group. End points due to flow monitoring in the group 1 (study group) and group 2 (placebo-treated control group) are compared to total end points

- Demonstrates the comparison of the mean AVFs' flow diagram (left columns) and the differences of arterial end-diastolic velocity (right columns) between the groups. In the early period, we detected high blood flows and lower arterial velocity in study group when we compared groups
No bleeding episode such as intracranial hemorrhage in gastrointestinal tract was recorded during the active treatment period. There were no differences between baseline and follow-up hematocrit values or changes in recombinant human erythropoietin doses during the study period for either group.
Discussion
In this prospective double-blind research, we presented our experiences of a new combination therapy and its efficacies on AVF access after surgery in our ESRD population. According to the study, clopidogrel in combination with oral prostacycline analog as an arterial vasodilator can be used to provide early AVF maturation in ESRDP with diabetes. According to previous reports, the size and quality of the arterial and venous access and distensibility after AVF creation play important roles. Therefore, we decided to use our combination therapy to provide more dilated and elastic arterial and venous access after AVF creation. Thus, blood flow from the fistula increased significantly in patients who were given a combination therapy than in patients who were given placebo. Our study findings support the importance of distensibility and quality of vessel access.
As we know that after AVF creation, anastomotic or juxta-anastomotic stenosis is the most common cause of a fistula failing to mature. The stenosis most likely occurs because of operative trauma. In case of transposed vein fistulas, the loss of vasa vasorum in the mobilized portion of the vein may also contribute to causation of stenosis. Second, there is the element of neo-intimal hyperplasia. Because of a large pressure gradient between arterial and venous circulation, blood accelerates while passing from the artery to the vein causing turbulent flow that is considered responsible for endothelial damage as a result of micro-trauma. In addition, unfortunately, some patients have a small arterial and/or venous access diameter. Therefore, effective hemodialysis can be impossible due to occlusion and/or stenosis of the venous or arterial line. It is shown that venous distensibility is a major problem after AVF creation. In Van der Linden et al.'s study, it was demonstrated that venous distensibility was significantly correlated with AVF maturation, especially in patients with vein diameter ≤2 mm.[12] After AVF creation, arterialization of the venous access starts in the future. Thus, if there is no suitable venous distensibility, AVF maturation and survival will be a problem. According to this hypothesis, we believe that the effective vasodilatation can be required in these particular groups for early AVF maturation and longer survival.
There have been a few small trials evaluating pharmacological agents aimed at reducing the early thrombosis rate.[8131415]. These trials utilized the antiplatelet agents, aspirin, sulphinpyrazone, and clopidogrel and varied in size from as few as 5 patients up to 250 patients. Andrassy et al. compared patients given aspirin for 4 weeks with placebo; the thrombosis rate in the aspirin group was 4% compared with the control group of 24%.[15] In another study, Kooistra et al. demonstrated that a low-dose aspirin could not prevent thrombovascular accidents in hemodialysis patients during treatment with recombinant human erythropoietin.[8] Therefore, they did not demonstrate any benefit from aspirin.
In Gröntoft et al.'s study, it was demonstrated that there was a statistical significance when compared to fistula thrombosis rates in clopidogrel groups versus placebo groups (12% vs. 19%).[16] Studies using sulphinpyrazone showed variable results, but were underpowered with the largest study enrolling only a small number of patients.[17] Pooled data for clopidogrel suggests a reduction in thrombosis rate from 25% to 12%. Thus, although there is some data to suggest that antiplatelet agents may increase the primary patency of AVF, the limited evidence base and the uncertainty regarding the choice of agent have not supported the widespread use of antiplatelet agents in the prevention of AVF thrombosis. However, for the first time, our study showed the effects of clopidogrel in combination with oral prostacycline agent on autogenous AVF creation.
We know that autogenous AVF is the preferred hemodialysis access and is associated with lower morbidity and mortality than AVGs. However, 20–60% of fistulas fail to mature sufficiently to support dialysis.[34618] Thus, the high rate of maturation failure is a major unsolved problem, yet.
The costs and morbidity problems due to AVF maturation failure among hemodialysis patients are still reported worldwide.[1920] Vascular access surgeons are faced with the same challenge all over the world – choosing the best vascular access type for chronic hemodialysis. Results of different studies are sometimes confounding. Size and quality of the vessels seem to be the important factors known. Despite the experiences of AVF in ESRDP, considerable amount of time is required for the fistula to mature and suitably develop into a functional format.[192021] A number of authors and Dember et al. have maintained that outflow venous stenosis and vascular intimal hyperplasia were the main causes of AVF failure.[456721] AVF thrombosis resulting from neointimal hyperplasia can be initiated by activated platelet function, endothelial cell injury, vascular smooth muscle cell proliferation, and unphysiologic vascular anastomosis.[81017] In view of these postulated mechanisms of AVF failure, possible clinical benefit of antiplatelet therapy has been proposed for prevention of AVF failure.[131416]
In particular, aspirin, the most widespread antiplatelet drug, has been evaluated as prophylaxis for AVF failure in several studies, mainly among the use of AVG.[19] In a study, the authors examined the efficacy of combination therapy of aspirin and clopidogrel for prevention of arteriovenous graft failure,[19] but this study was stopped before completion because the prevalence of hemorrhagic complication was significantly higher in the intervention group. In the study group, only 28% of patients required re-intervention during the 1-year follow-up period. However, 52% of placebo group needed re-intervention due to AVF failure.
During the aspirin therapy, the most important problem is hemorrhage in hemodialysis patients than in the general population because hemodialysis patients are more likely to have platelet dysfunction[8] and they receive intermittent anticoagulant therapy during hemodialysis sessions. However, clopidogrel is tolerated by the patient and is not a cause of gastrointestinal bleeding. Thrombosis or severe occlusion of autogenous AVF can be seen within the first several weeks following surgical creation, or inadequate maturation of the vein. AVF evaluation 4–6 weeks after creation should be considered mandatory.[222324]
In the literature, the primary AVF failure rate is approximately 9–50%.[222425] Some of the previous studies have indicated that the frequency of AVF failure can be reduced with antiplatelet agents; but in other studies the authors maintained antiplatelet therapy did not affect AVF maturation and survival.[2223] Kheda et al. reported that low arterial elasticity was associated with AVF stenosis and fistula maturation failure.[22] They also concluded that vessel dilation was not needed for adequate blood flow except at the smaller diameters. Therefore, they speculated that low elasticity promotes development of early stenosis of AVFs.
The successful results of our analysis suggest that to inhibit AVF failure, daily administration of 75 mg oral clopidogrel and arterial prostacycline analog administration may be initiated 7–10 days prior to AVF creation.
In contrary to our results, Yevzlin et al.'s trial suggested that the use of antiplatelet was not associated with significant risk reduction in access failure.[4] Kaufman demonstrated no change in the risk of graft thrombosis with aspirin plus clopidogrel therapy.[11] The author reported that in chronic hemodialysis patients there was a trend toward increased or did not change thrombosis rate of AVF using aspirin therapy.[11] Some surgeons choose the synthetic polytetrafluoroethylene graft for the creation of AVF. In Fiskerstrand et al.'s study, 2 out of 6 patients in the ticlopidine group compared with 5 out of 9 in the placebo group, developed fistulae thromboses in 1 month.[23] In the previously published study by Gröntoft et al., there were only 2 out of 19 patients who received treatment developed for fistulae thromboses compared to 8 out of 17 on placebo.[16] The results of adequacy for AVFs developed sufficiently to be used for dialysis in 46.5% of patients.[16] Miller et al. and Allon and Robbin have suggested that the higher adequacy rate could be provided in upper arm location and multidisciplinary team approach to vascular access by the surgeons, nurses, and radiologists.[56]
Our results showed that there are some differences from the previous studies. The arterial diameter, elasticity, and the venous line distensibility are the main factors of fistula maturation. In publications from DAC consortium, a large number of patients from different centers are included.[721] The authors did use only clopidogrel as an antiplatelet. Dember et al. followed the patients for a long time (approximately 4 years) but we could follow the cases for a year only. Arterial stiffness is the most important factor in diabetes. Our therapy model may be provided more distensible and large size venous line, and more elastic arterial access. Our study differences may be related to surgical experiences, patients' age, smoking history, BMI, patients' arterial and venous diameters, and distensibility.
Conclusion
Multiple individuals are involved in the management of vascular access, including nephrologists, access surgeons, radiologists, dialysis nurses, and the patient. Achieving optimal vascular access outcomes requires agreement on a common set of goals by all these individuals, close collaboration, and good communication. However, primary AVF failure remains a major problem for hemodialysis patients worldwide. According to our study, it is observed that prophylaxis of vascular access thrombosis needs to start earlier in the hemodialysis patients. Clopidogrel and oral prostacycline analog, which begins 7–10 days prior to AVF creation and continuing during follow-up period seems to be effective and safe, and inhibit primary AVF failure with acceptable side effects in hemodialysis patients.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Early arteriovenous fistula failure: A logical proposal for when and how to intervene. Clin J Am Soc Nephrol. 2006;1:332-9.
- [Google Scholar]
- Predictors of adequacy of arteriovenous fistulas in hemodialysis patients. Kidney Int. 1999;56:275-80.
- [Google Scholar]
- Increasing arteriovenous fistulas in hemodialysis patients: Problems and solutions. Kidney Int. 2002;62:1109-24.
- [Google Scholar]
- Effect of clopidogrel on early failure of arteriovenous fistulas for hemodialysis: A randomized controlled trial. JAMA. 2008;299:2164-71.
- [Google Scholar]
- A low dose aspirin does not prevent thrombovascular accidents in low-risk hemodialyses patients during treatment with recombinant human erythropoietin. Nephrol Dial transplant. 1994;9:1115-20.
- [Google Scholar]
- Retrospective analysis of 271 arteriovenous fistulas as vascular access for hemodialysis. Indian J Nephrol. 2013;23:191-5.
- [Google Scholar]
- Association between vascular access failure and the use of specific drugs: The Dialysis Outcomes and Practice Patterns Study (DOPPS) Am J Kidney Dis. 2002;40:1255-63.
- [Google Scholar]
- Antithrombotic agents and the prevention of access thrombosis. Semin Dial. 2000;13:40-6.
- [Google Scholar]
- Forearm venous distensibility predicts successful arteriovenous fistula. Am J Kidney Dis. 2006;47:1013-9.
- [Google Scholar]
- Randomized controlled trial of clopidogrel to prevent primary arteriovenous fistula failure in hemodialysis patients. Indian J Nephrol. 2009;19:57-61.
- [Google Scholar]
- Thromboprophylactic effect of ticlopidine in arteriovenous fistulas for haemodialysis. Scand J Urol Nephrol. 1985;19:55-7.
- [Google Scholar]
- Prevention of p.o. clotting of av. Cimino fistulae with acetylsalicyl acid: Results of a prospective double blind study. Klin Wochenschr. 1974;52:348-9.
- [Google Scholar]
- Effects of ticlopidine in AV-fistula surgery in uremia. Fistula Study Group. Scand J Urol Nephrol. 1998;32:276-83.
- [Google Scholar]
- Influence of antiplatelet drugs on occlusion of arteriovenous fistula in uraemic patients. Int Urol Nephrol. 1992;24:83-9.
- [Google Scholar]
- Acquired platelet dysfunction in patients with uremia. Hematol Oncol Clin North Am. 1990;4:129-43.
- [Google Scholar]
- Design of the Dialysis Access Consortium (DAC) Clopidogrel Prevention of Early AV Fistula Thrombosis Trial. Clin Trials. 2005;2:413-22.
- [Google Scholar]
- Anti-platelet therapy in graft thrombosis: Results of a prospective, randomized, double-blind study. Kidney Int. 1994;45:1477-83.
- [Google Scholar]
- Prevention of early thrombus formation in arteriovenous fistulae. Dial Transplant. 1981;10:167.
- [Google Scholar]
- Influence of arterial elasticity and vessel dilatation on arteriovenous fistula maturation: A prospective cohort study. Nephrol Dial Transplant. 2010;25:525-31.
- [Google Scholar]
- Double-blind randomized trial of the effect of ticlopidine in arteriovenous fistulas for hemodialysis. Artif Organs. 1985;9:61-3.
- [Google Scholar]
- Pharmacologic prevention of vascular access stenosis. Curr Opin Nephrol Hypertens. 1999;8:569-72.
- [Google Scholar]
- Hemodialysis vascular access dysfunction: From pathophysiology to novel therapies. Blood Purif. 2003;21:99-110.
- [Google Scholar]







