Translate this page into:
Induction with rATG versus No-induction in Deceased Donor Renal Transplantation – A Retrospective Observational Study
Address for correspondence: Dr. Sree Bhushan Raju, Professor, Deptartment of Nephrology, Nizams Institute of Medical Sciences, Hyderabad, Telangana, India. E-mail: sreebhushan@hotmail.com
-
Received: ,
Accepted: ,
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
This article was originally published by Wolters Kluwer - Medknow and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Introduction:
Deceased donor renal transplantation (DDRT) poses special immunological challenges; particularly in resource-poor scenarios. There is substantial evidence that rabbit antithymocyte globulin (rATG) is superior to interleukin-2 receptor blocker and placebo among patients at high immunological risk. However, due to the lack of randomized controlled trials, this remains controversial in DDRT maintained on tacrolimus/mycophenolic acid/steroids. Here, in this study, we compared the clinical outcomes of induction with rATG therapy to no-induction therapy.
Methods:
The study was a single-center, retrospective cohort study. A total of 62 patients were divided into two groups, based on induction immunosuppression; induction with rATG (N = 25) and no-induction group (N = 37). Both groups received tacrolimus/mycophenolate mofetil sodium/prednisolone as maintenance immunosuppression. The main outcomes were incidence of acute rejection (AR) within the first year and graft survival at the end of 1 year.
Results:
The AR at the end of 1-year was reported as 8% and 27% for the induction and no-induction groups (P = 0.07), respectively. A total of 15 patients died. Patient survival rates at 12 months were 83.8% (no-induction) and 64.0% (induction; P = 0.094). Death-censored graft survival rates, 12 months after transplantation, were similar in both treatment groups (83.7% vs. 83.5%, P = 0.972). The incidence of death with functioning graft was significantly high in the induction group (28% vs. 5.4%, P = 0.045).
Conclusion:
The incidence of AR was less in patients who received rATG induction compared with patients who did not receive any form of induction. An added advantage of induction with ATG in terms of reduced incidence of AR must be weighed against high incidence of infection, death with functioning graft, and death.
Keywords
Deceased donor
India
induction
rATG
renal transplantation
Introduction
Renal transplantation is an ideal treatment modality for end-stage renal disease; however, demand for kidneys exceeds supply in all parts of the world. Deceased donor renal transplantation (DDRT) is still infrequent in India, constituting less than 5% of the total renal transplants of about 3,500 per year.[1]There have been tremendous advances in general medical management both before and after renal transplantation over the past decades. The practice of combination therapy including induction and maintenance immunosuppression has become common. The primary reason for the use of induction therapy, or intense immunosuppressive therapy at the time of transplant, is to avoid early acute rejection (AR) historically known to predict late graft loss. The current Kidney Disease Improving Global Outcomes Work Group guidelines recommend interleukin-2 receptor antagonist (IL2-RA) as a first–line induction therapy across all types of donor–recipient profiles to reduce AR risk and allograft loss.[2] Also, there is substantial evidence that rabbit antithymocyte globulin (rATG) is superior to IL2-RA and placebo among patients at high immunological risk. However, in the era of tacrolimus/mycophenolic acid (Tac/MPA) maintenance regimens with or without steroids, controversy exists with respect to the added benefit of induction therapy on outcomes of renal transplantation.[3] There are recent data that support that no-induction-based regimen can achieve similar outcomes compared with induction-based regimen. The present study was designed to compare the short-term outcomes of DDRT between induction with rATG and no-induction group.
Methods
This study was a single-center, retrospective observational study, with prospective follow-up of adult patients aged >18 years who underwent DDRT between 2016 and 2019. Patients with age younger than 18 years old, multiorgan transplantations, two or more previous kidney transplantations, and recipients of induction agents other than ATG were excluded from the study. The research followed the Declaration of Helsinki and was approved by the institutional ethical committee (EC/NIMS/2433-2019). The primary objective of the study was the incidence of AR within the period of 12 months and overall graft failure at the end of 1 year after DDRT. The secondary objectives were immediate graft outcome and infection episodes at the end of 1 year.
Immunosuppressive regimen
The patients were started on Tac and mycophenolate sodium (MPS) (first dose just prior to transplantation). In the induction group, the patients received Tac 0.10 mg/kg/day in two divided doses, while in the no-induction group, the patients received Tac 0.12 mg/kg/day in two divided doses. All patients received MPS 720 mg twice a day. In both groups, the patients received intravenous (IV) methyl prednisolone 1,000 mg/day for 3 days, with the first dose being started during intraoperative period. rATG was given as 1mg/kg for 3 days with cumulative dose of 3 mg/kg, and the first dose was given during the intraoperative period. The Tac level was targeted based on institute protocol. The Tac level during hospital stay was measured on Day 1 and Day 5 while regular monitoring was done for desired therapeutic level. For the first 3-month target, the Tac level was 10–15 ng/mL in the no-induction group, where as 5–10 ng/mL in rATG group >3–12-month target Tac level was 5–10 in both the induction and the no-induction groups. Tac dose reduction was done based on desired therapeutic level. Tac level was also measured during episodes of rejection. Tac dose was altered during the episodes of rejection if deemed necessary. The patients were discharged with triple immunosuppression including prednisolone, Tac, and MPS. Patients in both groups received cotrimoxazole prophylaxis (80/400 mg once daily for 12 months). Patients in the rATG group received valganciclovir prophylaxis as well (valganciclovir 450 mg once daily for 200 days).
Immediate graft function was defined as nonrequirement of dialysis after transplantation and nadir serum creatinine reached within normal limits at the time of discharge. Delayed graft function (DGF) was defined as the need for dialytic support within a week of transplant except for hyperkalemia. All AR were biopsy-proven. A standard protocol was used for rejection management. All patients received methyl prednisolone, and in case of no response, rATG and PLEX + IVIG were given. Overall allograft failure was defined as a return to dialysis, retransplant, or death with functioning allograft after transplantation was included, till the end of the last follow-up date. Patient survival was calculated from the date of transplantation to the date of death or the date of the last follow-up. Graft survival (non-censored for death) was calculated from the date of transplantation to the date of irreversible graft failure signified by return to long-term dialysis (or re-transplantation) or the date of the last follow-up during the period when the transplant was functioning or to the date of death. Here, death with graft function was treated as graft failure. In the event of death with a functioning graft (DWFG), the follow-up period was censored at the date of death.
Statistical analysis
Statistical software SPSS (Statistical Package for the Social Sciences) Version 22.0 (SPSS, Chicago, IL, USA) was used for statistical analysis. Quantitative data were expressed as mean ± standard deviation for normally distributed data or median with 25% and 75% interquartile range for nonnormally distributed data. Categorical data were expressed as percentages. For comparison of clinical and pathological features of patients, the Student's t-test, one-way ANOVA (analysis of variance), Chi-square test, and Mann–Whitney U tests were used. Kaplan–Meier curves were used to analyze the patients' survival and graft survival. Survival analysis was performed using the log-rank test. Multivariate analysis with Cox regression was used to determine the prognostic factors. Statistical significance was considered as P < 0.05.
Results
In the final analysis, a total of 62 patients were included, out of which 25 received induction (steroid + ATG) and 37 patients received no-induction (steroid only). Figure 1 depicts flow diagram of the study. A few patients initially could not be given total dose and a few withdrew from the protocol due to financial reasons, and a few were deleted due to insufficient data as it is a retrospective analysis. A few were lost to follow-up to some other hospital due to practical issues. This led to the discrepancy in the number of patients analyzed in each group.

- Flow diagram of the study
Tables 1 and 2 describe the baseline demographics and clinical and laboratory characteristics of the cohort respectively. No significant difference was observed between the two groups. The mean age of the patients was 37.48 ± 11.34 years, with male preponderance (64.5%). The most common native kidney disease in recipients was chronic glomerulonephritis (35.4%) followed by chronic interstitial nephritis (32.3%) and diabetic kidney disease (6.5%).
| Characteristics | Overall | No-induction (Steroid only) | Induction (Steroid + ATG) | P |
|---|---|---|---|---|
| n | 62 | 37 | 25 | |
| Age (years) | 37.48±11.34 | 38.46±11.81 | 36.04±10.65 | 0.414 |
| Gender | 0.914 | |||
| Male (%) | 40 (64.5%) | 24 (64.9%) | 16 (64.0%) | |
| Body Mass Index (kg/m2) | 22.34±3.72 | 22.64±3.49 | 21.89±4.06 | 0.439 |
| Smokers (%) | 11 (17.7%) | 7 (18.9%) | 4 (16.0%) | 0.768 |
| ABO Blood Group | ||||
| O | 33 (53.2%) | 21 (56.8%) | 12 (48.0%) | |
| A | 13 (21.0%) | 8 (21.6%) | 5 (20.0%) | |
| B | 13 (21.0%) | 6 (16.2%) | 7 (28.0%) | |
| AB | 3 (4.8%) | 2 (5.4%) | 1 (4.0%) | |
| Native Kidney Disease | 0.203 | |||
| Chronic Glomerulonephritis | 22 (35.4%) | 15 (40.5%) | 7 (28.0%) | |
| Chronic Interstitial Nephritis | 20 (32.3%) | 7 (18.9%) | 13 (52.0%) | |
| Diabetic Kidney Disease | 5 (8.1%) | 4 (10.8%) | 1 (4.0%) | |
| Hypertensive Nephropathy | 6 (9.7%) | 4 (10.8%) | 2 (8.0%) | |
| Polycystic Kidney Disease | 4 (6.5%) | 3 (8.1%) | 1 (4.0%) | |
| Others | 5 (8.1%) | 4 (10.8%) | 1 (4.0%) | |
| Diabetes | 8 (12.9%) | 4 (10.8%) | 4 (16.0%) | 0.550 |
| Hypertension | 59 (95.2%) | 35 (94.6%) | 24 (96.0%) | 0.800 |
| Cardiovascular Disease | 12 (19.4%) | 7 (18.9%) | 5 (20.0%) | 0.916 |
| Dialysis Type | 0.231 | |||
| Hemodialysis | 53 (85.5%) | 30 (81.1%) | 23 (92.0%) | |
| Peritoneal Dialysis | 9 (14.5%) | 7 (18.9%) | 2 (8.0%) | |
| Dialysis Duration Before Transplantation (years) | 0.136 | |||
| <3 | 33 (53.2%) | 20 (54.1%) | 13 (52.0%) | |
| 3.5 | 18 (29.0%) | 8 (21.6%) | 10 (40.0%) | |
| >5 | 11 (17.7%) | 9 (24.3%) | 2 (8.0%) | |
| Residual Urine Output/Day (mL) | 0.800 | |||
| ≤100 | 26 (41.9%) | 16 (43.2%) | 10 (40.0%) | |
| >100 | 36 (58.1%) | 21 (56.8%) | 15 (60.0%) | |
| HCV seroconversion | 13 (21.0%) | 9 (24.3%) | 4 (16%) | 0.430 |
| Sensitization History | ||||
| Blood transfusion (%) | 51 (82.3%) | 31 (83.8%) | 20 (80.0%) | 0.702 |
| Pregnancy (%) | 17 (81.0%) | 11 (84.6%) | 6 (75.0%) | 0.586 |
ATG=antithymocyte globulin; HCV=hepatitis C virus
| Characteristics | Overall | No-induction (Steroid only) | Induction (Steroid + ATG) | P |
|---|---|---|---|---|
| Age (years) | 37.74±15.25 | 41±15.34 | 32.88±14.05 | 0.038 |
| Gender | ||||
| Male (%) | 48 (77.4%) | 26 (70.3%) | 22 (88.0%) | 0.101 |
| BMI (kg/m2) | 25.18±4.03 | 24.84±4.12 | 25.39±3.92 | 0.424 |
| ABO Blood Group | ||||
| O | 31 (50.0) | 20 (54.1%) | 11 (44.0%) | |
| A | 13 (21.0%) | 8 (21.6%) | 5 (20.0%) | |
| B | 15 (24.2%) | 7 (18.9%) | 8 (32.0%) | |
| AB | 3 (4.8%) | 2 (5.4%) | 1 (4.0%) | |
| Terminal Creatinine (mg/dL) | 1.13±0.513 | 1.13±0.55 | 1.14±0.47 | 0.912 |
| Comorbidities | ||||
| Hypertension (%) | 13 (21.0%) | 10 (27.0%) | 3 (12.0%) | 0.154 |
| Diabetes (%) | 5 (8.1%) | 5 (13.5%) | 0 (0.0%) | 0.055 |
| Cause of Death | 0.241 | |||
| CVA | 16 (25.8%) | 12 (32.4%) | 4 (16.0%) | |
| Head Trauma | 44 (71.0%) | 24 (64.9%) | 20 (80.0%) | |
| ECD (%) | 11 (17.7%) | 8 (21.6%) | 3 (12.0%) | 0.331 |
| KDPI (%) | 41.62±26.59 | 45.97±26.73 | 35.2±25.54 | 0.118 |
| KDRI | 0.967±0.306 | 1.01±0.32 | 0.89±0.26 | 0.121 |
| Cold Ischemia Time | 6.69±2.24 | 6.56±2.049 | 8.48±7.048 | 0.126 |
| Weight D/R ratio | 1.16±0.27 | 1.14±0.28 | 1.21±0.27 | 0.372 |
| Acute Kidney Injury (%) | 14 (22.6%) | 8 (21.6%) | 6 (24.0%) | 0.827 |
ATG=antithymocyte globulin; BMI=body mass index; CVA=cerebrovascular accident; ECD=extended criteria donor, KDPI=Kidney Donor Profile Index; KDRI=Kidney Donor Risk Index; D/R=donor-recipient
On an average, the patients in the induction group received rATG 142.6 ± 40.10 mg over 3 to 5 days. rATG was prematurely stopped in one due to sepsis, and the dose was reduced by 50% due to leukopenia in two. [Table 3] depicts Graft and patient outcome at the end of 1 year
| Type | Overall | No-induction (Steroid only) | Induction (Steroid + ATG) | P |
|---|---|---|---|---|
| Mean e-GFR mL/min (CKD-EPI) | ||||
| 6 months | 81.15±25.99 | 73.28±30.30 | 0.338 | |
| 12 months | 80.37±25.51 | 77.81±34.16 | 0.777 | |
| Delayed graft function | 26/62 (41.9%) | 17/37 (45.9%) | 9/25 (36.0%) | 0.4402 |
| Acute rejection episodes (%) | 12/62 (19.4%) | 10/37 (27.0%) | 2/25 (8.0%) | 0.063 |
| Overall allograft failure at 12 months (%) | 17/62 (27.4%) | 8/37 (21.6%) | 9/25 (36%) | 0.2168 |
| Death with functioning allograft at 12 months n/(%) | 9/62 (14.5%) | 2/37 (5.4%) | 7/25 (28.0%) | 0.045 |
| Death (%) | 15/62 (24.2%) | 6/37 (16.2%) | 9/25 (36.0%) | 0.074 |
| PTDM | 12/62 (19.4%) | 8/37 (21.6%) | 4/25 (16%) | 0.58 |
| Anemia | 20/62 (32.3%) | 12/37 (18.9%) | 8/25 (32.0%) | 0.241 |
| Leukopenia | 18/62 (29.0%) | 10/37 (27.0%) | 8/25 (32.0%) | 0.674 |
| Thrombocytopenia | 15/62 (24.2%) | 9/37 (24.3%) | 6/25 (24.0%) | 0.9772 |
| UTI | 22/62 (35.5%) | 11/37 (29.7%) | 11/25 (44.0%) | 0.25 |
| Pneumonia | 12/62 (19.4%) | 6/37 (16.2%) | 6/25 (24.0%) | 0.449 |
| PCP | 2/62 (3.2%) | 1/37 (2.7%) | 1/25 (4.0%) | 0.7780 |
| Sepsis | 10/62 (16.1%) | 4/37 (10.8%) | 6/25 (24.0%) | 0.169 |
ATG=antithymocyte globulin; e-GFR=estimated glomerular filtration rate; CKD-EPI=chronic kidney disease-epidemiology; PTDM=post-transplant diabetes mellitus; UTI=urinary tract infection; PCP=PNEUMOCYSTIS pneumonia
Only two patients (8.0%) experienced biopsy-proven AR (BPAR) in the induction group compared with10 patients (27.0%) in the no-induction group; however, this difference was statistically nonsignificant (P = 0.063). In the no-induction group, among 10 episodes of BPAR, five out of seven early rejections responded to antirejection therapy. However, it was not analyzed as the cases were either not biopsy-proven and responded to one or two doses of therapy with methyl prednisolone. There were three late rejections in this group. Only one out of three of them showed improvement with antirejection therapy where as two out of two rejections were late rejections in the induction group, and both responded to antirejection therapy. During the remaining study period (Day 30–365), 3/10 (30%) of the rejections in the no-induction group and 2/2 (100%) in the induction group were reported. Out of 12 BPAR, three were acute cellular rejections and nine were antibody-mediated rejection. The results of covariate-adjusted multivariate logistic regression models for AR within 1-year posttransplant compared with the no-induction group are shown in Table 4. There was no significant difference between the two groups. In both treatment groups, graft function was achieved rapidly. Overall, DGF was 41.9% in the study for the most frequent adverse events, “urinary tract infection” (UTI) (induction 44% vs. no-induction 29.7%) and “anemia” (32.0% vs. 18.9%). The incidence of cytomegalo virus infection was more in the no-induction group (45.9% vs. 24%). Posttransplant diabetes mellitus (PTDM) was observed in 19.4% of patients.
| Induction group | Overall graft failure within 1 year of transplant (%) | Adjusted* HR$ | 95% CI | P |
|---|---|---|---|---|
| Induction group (Steroid + ATG) | 36.0% | 1.691 | 0.651-4.38 | 0.2804 |
| No-induction group (Steroid only) | 21.6% | |||
| $HR=Hazards ratio; 95% CI=95% confidence interval. *Adjusted for donor factors (age, sex and kidney donor profile index, cold ischemia time, and donor to recipient weight ratio) and recipient factors (age, sex, diabetes status, Hypertension, cardiovascular disease, and dialysis vintage) | ||||
| Induction group | Acute rejection rate within 1 year (%) | Adjusted* OR$ | 95% CI | P |
| Induction group (Steroid + ATG) | 8.0% | 0.208 | 0.038-1.136 | 0.070 |
| No-induction group (Steroid only) | 27.0% | |||
$OR=Odds ratio; 95% CI=95% confidence interval. *Adjusted for donor factors (age, sex and kidney donor profile index, cold ischemia time, and donor to recipient weight ratio) and recipient factors (age, sex, diabetes status, hypertension, cardiovascular disease, and dialysis vintage). ATG=antithymocyte globulin
Discussion
We have observed an overall incidence of AR up to 19.4%, and in the induction with rATG group it was 8% compared with the no-induction group with 27% (P = 0.063); this was comparable with other Indian studies.[456]A randomized, double-blinded study compared AR, graft failure, and death in a cohort of 386 adult renal transplant recipients (DDRT comprising 42% of the cohort) who received antibody induction (IL2-RA vs. rATG), Tac/MPA maintenance immunosuppression, and steroid cessation within 7 days with those receiving chronic low-dose steroids.[7]There was no difference in primary end points (composite of death, graft loss, or moderate/severe AR) at 5 years, but subgroup analysis showed a significantly higher rate of BPAR in recipients induced with IL2-RA (24.4%) compared with rATG (14.4%) in the no-steroid group. Exposure to immunosuppressive medication was similar in both groups, and the recommended dosages and trough-level ranges were followed. Whole-blood Tac trough levels remained within the target ranges and were similar to those observed in other studies. The rejections in the induction group occurred later than in the no-induction group, in which the highest incidence of rejection was observed in the first 2 weeks after transplantation. Although the difference in rejection rates was not statistically significant, the graft survival did not appreciably differ between the treatment groups 12 months after transplantation; thus, even in patients who had a rejection, this did not lead to graft loss. However, as AR is the most important risk factor for chronic rejection, further long-term follow-up is required. When multivariate logistic regression analysis was applied, though, odds of rejection were less in the induction group, but it was statistically nonsignificant. In a study done by Tanriover et al.,[8] where they studied the United Network for Organ Sharing Registry for patients receiving DDRT from 2000 to 2012 maintained on Tac/MPA at transplantation hospital discharge (n = 74,627) to compare outcomes of IL2-RA and other induction agents. They found that AR at 1 year was significantly lower with ATG (9.1% vs. 13.3%).
In the current study, no significant difference in the incidence of CMV infection between the induction and no-induction group (P = 0.08) was detected. Yang et al.[9] and Hardinger et al.[10] reported no significant difference in CMV infection in comparison of two doses of thymoglobulin. In contrast to these studies, Castro et al.,[11] who compared two dosages of thymoglobulin, reported a higher incidence of CMV infection in higher dosage, whereas the rate of this infection was 33% in the study. In fact, the incidence was numerically low in induction with the ATG group, which might be due to primary CMV prophylaxis in all ATG-receiving candidates. Bacterial infections are the most common form of infections reported after rATG induction therapy. Multiple cofactors are generally present, including technical complications from surgery, urinary and vascular catheters, and complex immunosuppressive regimens. In our study, 30.6% of patients had bacterial infection, and the most common form of presentation was UTI followed by pneumonia and sepsis. There was no significant difference between the two groups. In the study done by Mourad et al.,[12] ATG was not associated with an increased risk of bacterial infection, compared with the risk with no-induction therapy. Cytopenia was expected in the induction group due to the ATG effect on blood cells. The most common hematological complication in our study was anemia (32.3%) followed by leukopenia (29.0%) and thrombocytopenia (24.2%). However, there was no significant difference between the two groups and similar results have been reported in other studies.[813]
In India, burden of infectious disease is high, which poses a special challenge in immunocompromised patients. The argument behind use of induction agent is essentially to avoid DGF and AR. In our study also, the incidence of DGF and AR was numerically low in induction group, but graft survival rate was more in the non-induction group.
Death-censored graft survival at 1 year between the two groups was similar (83.7% vs. 83.5%) in our study, and there was no significant difference between the groups, which is like previous studies.[1213] The most common cause of graft loss in our study was death (53%), and the most common cause for graft loss was sepsis followed by refractory rejection. Mouradet al.[12] have reported similar results. DWFG at 1 year in our study was 14.5%, which is like other previous studies that reported between 9% and38%. The most common cause of DWFG was sepsis. DWFG was significantly high in the induction group. Contrary to our study, Mourad et al.[12] reported similar incidence of DWGF between the ATG induction and non-induction groups. As sepsis was the most common cause of DWFG, long-term immunosuppression by ATG affecting both innate and acquired immunity might be one of the contributing factors.[141516]Patient survival rates at1 year were similar in both the groups – that is, 83.8% (no-induction) and 64.0% (induction; P = 0.094). The overall patient survival in our study participants was 75.8%, which implies high mortality [Figure 2]. The survival rate was affected by the two patients who died in the third and fifth day of transplant.
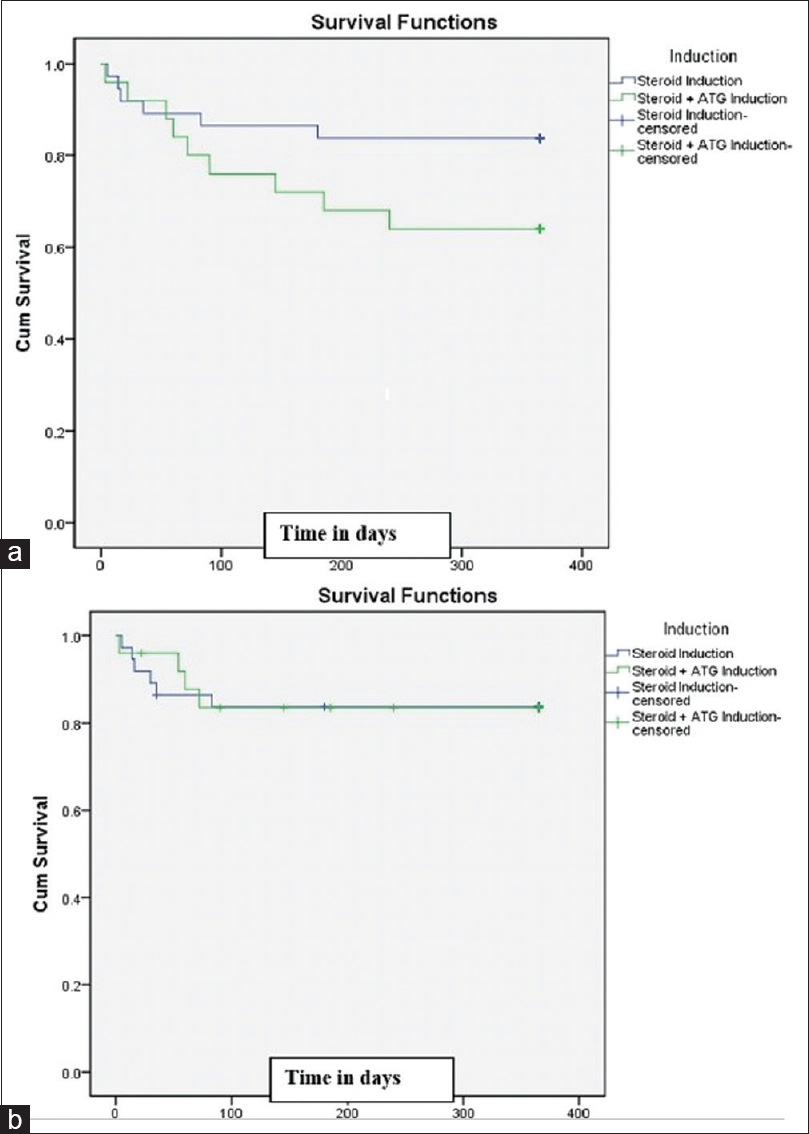
- Kaplan–Meier curve for (a) Patient survival at 12 months (P = 0.094);(b) Death-censored graft survival at 12 months (P = 0.972)
Limitations of the study
This was a single-center study, so results cannot be generalized to other population. Other factors such as small sample size, retrospective study design, and recipient's socioeconomic status might have affected the outcome of the study. The impact of human leukocyte antigen matching, panel reactive antibody, and donor-specific antibody could not be accessed due to logistic reasons.
Conclusion
In the present study, the incidence of AR was less in patients who received rATG induction compared with patients who did not receive any form of induction. The most common cause of graft loss was DWFG, which was significantly high in the patients who received induction with rATG. Death-censored graft survival was comparable between the two groups. The added advantage of induction with ATG in terms of reduced incidence of AR must be weighed against high incidence of infection, DWFG, and mortality. A larger group of patients with longer duration of follow-up is required to evaluate this observation.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant. 2009;9(Suppl 3):S1-S155.
- [Google Scholar]
- A prospective, randomized, multicenter study evaluating early corticosteroid withdrawal with Thymoglobulin in living-donor kidney transplantation. Clin Transplant. 2010;24:73-83.
- [Google Scholar]
- Deceased donor renal transplantation: A single center experience. Indian J Nephrol. 2017;27:4-8.
- [Google Scholar]
- Outcome of live and deceased donor renal transplantation in patients aged ≥55 years: A single-center experience. Indian J Nephrol. 2014;24:9-14.
- [Google Scholar]
- Deceased donor organ transplantation with expanded criteria donors: A single-center experience from India. Transplant Proc. 2010;42:171-4.
- [Google Scholar]
- A prospective, randomized, double-blind, placebo-controlled multicenter trial comparing early (7 day) corticosteroid cessation versus long-term, low-dose corticosteroid therapy. Trans Meet Am SurgAssoc Am SurgAssoc. 2008;126:205-20.
- [Google Scholar]
- Induction therapies in live donor kidney transplantation on tacrolimus and mycophenolate with or without steroid maintenance. Clin J Am Soc Nephrol. 2015;10:1041-9.
- [Google Scholar]
- Comparison of clinical outcome of low-dose and high-dose rabbit antithymocyte globulin induction therapy in renal transplantation: A single-center experience. Ann Transplant. 2014;19:277-82.
- [Google Scholar]
- Thymoglobulin induction is safe and effective in live-donor renal transplantation: A single center experience. Transplantation. 2006;81:1285-9.
- [Google Scholar]
- Induction versus noninduction therapy in kidney transplantation: Considering different PRA levels and different induction therapies. Transplant Proc. 2004;36:874-6.
- [Google Scholar]
- Induction versus noninduction in renal transplant recipients with tacrolimus-based immunosuppression. Transplantation. 2001;72:1050-5.
- [Google Scholar]
- Comparison of clinical outcome of induction immunosuppressive therapy with thymoglobulin and standard therapy in kidney transplantation; A randomized double-blind clinical trial. J Nephropathol. 2020;9:e08.
- [Google Scholar]
- Rabbit antithymocyte globulin as induction immunotherapy for pediatric deceased donor kidney transplantation. J Urol. 2005;174:703-7.
- [Google Scholar]
- A randomized trial of three renal transplant induction antibodies: Early comparison of tacrolimus, mycophenolate mofetil, and steroid dosing, and newer immune-monitoring. Transplantation. 2005;80:457-65.
- [Google Scholar]
- A three arm study comparing immediate tacrolimus therapy with ATG induction therapy followed by either tacrolimus or cyclosporine in adult renal transplant recipients. TransplantProc. 2002;34:1625-6.
- [Google Scholar]







