Translate this page into:
Prevalence of lower urinary tract infection in South Indian type 2 diabetic subjects
Address for correspondence: Dr. Vijay Viswanathan, M.V. Hospital for Diabetes and Diabetes Research Centre, WHO Collaborating Centre for Research, Education and Training in Diabetes, No. 4, Main Road, Royapuram, Chennai - 600 013, India. E-mail: dr_vijay@vsnl.com
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications and was migrated to Scientific Scholar after the change of Publisher.
Abstract
This study was done to determine the prevalence of lower urinary tract infection (UTI), the causative pathogens, their antimicrobial pattern, and the recurrence of infection in type 2 diabetic subjects. A total of 1157 (M: F 428: 729) type 2 diabetic subjects were selected for this study. Midstream urine specimens were collected and the culture tests were done by a quantitative method whereas antimicrobial sensitivity was determined by using the Kirby-Bauer method. A significant colony count was seen in 495 (42.8%) subjects and an insignificant count in 350 (30.3%) subjects; there were a few cases of recurrent UTI. Women (47.9%) had a significantly higher prevalence of UTI than men (34.1%) (χ2 = 20.3, P < 0.0001). Except for BMI, UTI was significantly associated with age, duration of diabetes, and poor glycemic control in both sexes. About 533 pathogens of gram positive and gram negative bacilli were isolated from 495 subjects in this study. Escherichea coli (E. coli) was the most commonly found organism. Gram negative pathogens were found to be highly sensitive to sulbactum / cefoperazone and piperacillin / tazobactum. The prevalence of UTI was significantly higher in women than men with E. coli being the major isolated pathogen. Gram negative pathogens were highly sensitive to sulbactum / cefoperazone and piperacillin / tazobactum.
Keywords
Antimicrobial pattern
causative pathogens
prevalence
type 2 diabetes
lower urinary tract infection
Introduction
An association between urinary tract infection (UTI) and diabetes mellitus was noted in an autopsy series reported in the 1940s.[1] The urinary tract is the principal site of infection in diabetes. Changes in host defence mechanisms, the presence of diabetic cystopathy and of microvascular disease in the kidneys may play a role in the higher incidence of UTI in diabetic patients.[2] Urinary tract infections are the most commonly found bacterial infections, accounting for nearly seven million office visits and one million emergency department visits, resulting in 100,000 hospitalizations of women, the elderly, and patients with spinal cord injuries and/or catheters, multiple sclerosis, HIV, and also diabetes.[3]
Several severe and less commonly encountered UTIs are thought to occur more frequently in diabetic patients.[4] In a recent study from Europe, asymptomatic bacteriuria was more prevalent among women with diabetes (26%) than in women without diabetes (6%).[5] Diabetic patients are at a high risk of development of UTIs, so it is recommended that special attention is paid to them, especially for the management of bacterial UTIs.[6] Various risk factors such as sexual intercourse, age, duration of diabetes, glycemic control, and complications of diabetes are associated with UTI.[7]
Antimicrobial therapy should be guided both by in vitro sensitivity and clinical response. Asymptomatic bacteriuria in excess of 100,000 microorganisms/mL is also an indication for treatment. In either case, a 7–10 day course of the appropriate drug should eradicate the infection although this must be confirmed by re-culture.[8]
The aims of this study were to determine the prevalence of lower urinary tract infection, the causative pathogens, their antimicrobial pattern, and the recurrence in type 2 diabetic subjects.
Materials and Methods
A total of 1157 (M: F 428: 729) consecutive type 2 diabetic subjects were studied during a period of one year. Demography, anthropometry, and the duration of diabetes were recorded and the body mass index (BMI) (kg/m2) was calculated using height and weight measurements. Diagnosis of diabetes was made based on the WHO criteria.[9] Subjects who received antimicrobial drugs during the past one month, pregnant women, and those with involvement of upper tract and renal failure were excluded from the study. The Ethics committee of the institution approved the study and written informed consent was obtained from all the study subjects.
Midstream urine samples were collected from the patients after giving proper guidelines. The urine samples were immediately transported to the microbiology department. If the urine specimen was found to be contaminated with normal flora of the vagina and urethra, the subject was asked to submit another sample for analysis.
Samples were processed using the following standard microbiological procedures: Smears for Gram's staining,[10] culture for morphology, biochemical tests for identifying the species of the pathogens, and antimicrobial sensitivity by the Kirby-Bauer Method.[11] Quality control procedures were incorporated to assure the quality of the stains, media, biochemicals, and antibiotic discs.
A diagnosis of UTI was made if the urine cultures had >103 to >105 colony forming units (CFUs)/mL of a single potential pathogen or two potential pathogens. A pure culture of Staphylococcus aureus was considered to be significant regardless of the number of CFUs. The presence of yeast in any number was also considered to be significant.[12]
Glycosylated hemoglobin (HbA1C%) was estimated by an immunoturbidimetric method using the Hitachi 917 autoanalyzer. The body mass index and HbA1C% were available for a subsample (n = 400).
Statistical analysis
Data were expressed as mean, SD, and percentages. Student's ‘t’ test, Z test, and Chi-square test were used as required; P < 0.05 was considered significant. Analysis was performed using statistical package SPSS version 10.0 (SPSS, USA).
Results
Four hundred ninety-five (42.8%) subjects showed a significant colony count whereas 350 (30.3%) subjects had an insignificant colony count. No growth was seen in 281 (24.3%) specimens; there were 31 (2.7%) improperly collected specimens (if the colony count was >105 CFU/mL with three different organisms). Polymicrobial urinary tract infections were seen in 34 (2.9%) cases and 533 pathogens were isolated among the patients who had UTI. Symptomatic UTI was noted in 298 (60%) subjects.
Table 1 shows the genderwise prevalence of urinary tract infection. Women (47.9%) had a significantly higher prevalence of urinary tract infection than men (34.1%) (χ2 = 20.3, P < 0.0001). A total of 108 (21.8%) subjects were treated with oral hypoglycemic agents, 172 (34.7%) with insulin while the rest required combination therapy. The HbAC of those with and without were 9.5±2.2 and 8.1±1.9 respectively (P<0.0001).
| N | % | Men vs women Chi square, P value | |
|---|---|---|---|
| Total (n = 1157) | 495 | 42.8 | |
| Men (n = 428) | 146 | 34.1 | 20.3, < 0.0001 |
| Women (n = 729) | 349 | 47.9 |
Table 2 shows the clinical characteristics of patients with UTI. The prevalence of UTI was not very marked until the age of 45 years and thereafter, a significant increase was seen in both men and women. We found that the rates of the incidence of UTI increased with the increasing duration of diabetes.
| Clinical characteristics | Men n = 146 | Women n = 349 |
|---|---|---|
| Age (years) | ||
| <45 (n = 46) | 12 (8.2) | 34 (9.7) |
| 45–55 (n = 137) | 35 (23.9) | 102 (29.2) |
| >55 (n = 312) | 99 (67.8) | 213 (61) |
| χ2 = 116.6, | χ2 = 206.6, | |
| P < 0.0001 | P < 0.0001 | |
| Duration of diabetes (years) | ||
| <10 (n = 209) | 61 (41.8) | 148 (42.4) |
| ≥10 (n= 286) | 85 (58.2) | 201 (57.6) |
| Z = 1.8, | Z = 2.69, | |
| P= 0.07 | P= 0.007 | |
| BMI (kg/m2)* | (n = 114) | (n = 286) |
| <25 (n = 152) | 59 (51.7) | 93 (32.5) |
| ≥25 (n = 248) | 55 (48.2) | 193 (67.5) |
| Z = 0.18, | Z = 5.5, | |
| P = 0.85 | P < 0.001 | |
| HbA1c(%) | ||
| <8 (n = 85) | 22 (19.3) | 63 (22) |
| 8–9 (n = 64) | 15 (13.2) | 49 (17.1) |
| ≥9 (n = 251) | 74 (64.9) | 177 (61.9) |
| χ2 = 54.1, | χ2 = 101.7, | |
| P < 0.0001 | P < 0.0001 |
The percentage of patients with UTI varied among the nonobese (BMI < 25 kg/m2) and obese (BMI ≥ 25 kg/m2) women (Z = 5.5, P < 0.001) whereas it did not differ in men. Significant differences were noted between men and women. (Chi2 = 11.9, P = 0.0005). Poor glycemic control was significantly associated with UTI in both sexes. Age, duration of diabetes, and glycemic control did not show any significant differences between men and women.
About 533 pathogens were isolated from 495 subjects with UTI, out of which, 362 were gram negative bacilli, 100 were gram positive cocci, and 71 were of the Candida spp.
Figure 1, panel a shows the percentage-wise distribution of gram negative bacilli in which 258 (71.3%) of the patients had E. coli, 49 patients (13.5%) had Klebsiella spp., and 32 patients (8.8%) had Pseudomonas spp. Enterobacter spp. and Citrobacter spp. were present in only 2% of the gram negative bacilli-infected cases. Nonfermenting gram negative bacilli and Proteus spp. both were found only in 1% of the patients.
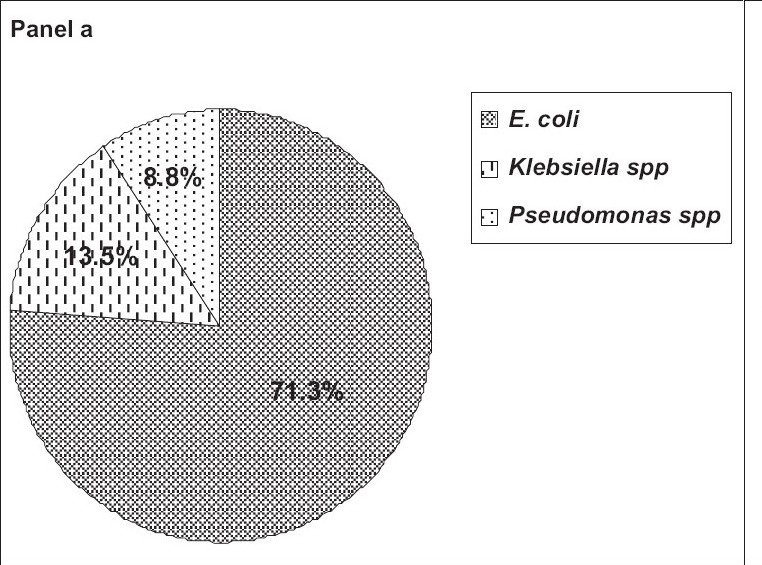
- Panel a shows the percentage-wise distribution of gram negative bacilli among UTI subjects
Figure 1, panel b shows the percentage-wise distribution of gram positive cocci isolated from infected cases of both sexes. About 59% had Enterococci spp. followed by coagulase-negative Staphylococcus (25%). Beta-hemolytic Streptococci were isolated from 8% of the patients. Nonhemolytic Streptococcus was found in 6% and Staphylococcus aureus was seen in 2% of the patients.
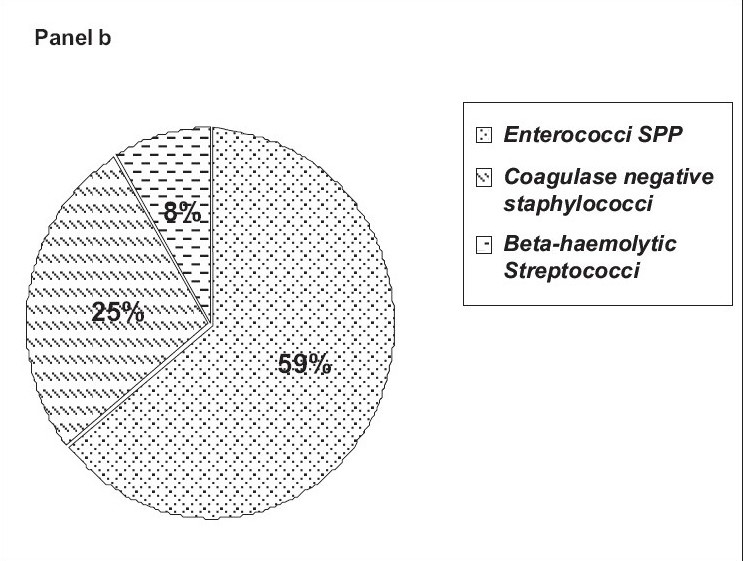
- Panel b shows the percentage-wise distribution of gram positive cocci among UTI subjects
Among the specimens containing Candida, 57 (80.3%) were Candida spp. and 14 (19.7%) were Candida albicans.
Table 3 shows the antimicrobial pattern of both gram negative bacilli and gram positive cocci. Gram negative bacilli were found to be highly sensitive to sulbactum / cefoperazone (91%) and piperacillin / tazobactum (83%). Gram positive cocci were 75% sensitive to sulbactum / cefoperazone and 66% sensitive to piperacillin / tazobactum respectively.
| Antimicrobials | Gram positive cocci (n = 100) values are in percentages | Gram negative bacilli (n = 362) |
|---|---|---|
| Amikacin | 29 | 65 |
| Netillin | 42 | 67 |
| Tobramycin | 14 | 30 |
| Sulbactum / cefoperazone | 75 | 91 |
| Pipercillin / tazobactum | 66 | 83 |
| Ciprofloxacin | 33 | 62 |
| Ofloxacin | 50 | 23 |
| Norfoxacin | 15 | 20 |
| Cefoperazone | 35 | 33 |
| Ceftzoxime | 48 | 62 |
| Cefotaxime | 63 | 51 |
Gram negative bacilli were found to be more sensitive than gram positive cocci to aminoglycosides such as netillin (67 vs 42%), amikacin (65 vs 29%), and Tobramycin (30 vs 14%).
Gram positive cocci (50%) were found to be more sensitive to ofloxacin than gram negative bacilli (23%) whereas gram negative bacilli (62%) were more sensitive than gram positive cocci (33%) to ciprofloxacin.
Not much difference in sensitivity was observed between gram positive cocci (35%) and gram negative bacilli (33%) to cefoperazone. Gram positive cocci (63%) were found to be more sensitive to cefotaxime than gram negative bacilli (51%), whereas gram negative bacilli (62%) were more sensitive than gram positive cocci (48%) to ceftizoxime.
Discussion
In this study, we found that the prevalence of lower UTI was significantly higher in female than in male type 2 diabetic patients. Evidence from various epidemiological studies showed that UTI is more common in women with diabetes than those without diabetes.[13] The high level of infection in the urinary tract of diabetic women may be determined by the number of microorganisms located in the vagina.[14]
UTI appears to be multifactorial in subjects with diabetes and various diabetes-related risk factors have been proposed. We observed that age, longer duration of diabetes, and poor glycemic control were significantly associated with UTI among subjects with diabetes. The presence of UTI varied among nonobese and obese subjects. A study from the Netherlands showed that among women with diabetes, older age, proteinuria, a lower body mass index, and a history of UTI were important risk factors for UTI.[5]
In another study, the longer duration of diabetes but not glucose control, was associated with the prevalence of bacteriuria.[15] The association between glycemic control and UTI among diabetic patients is controversial. In our study, only a few cases showed recurrent urinary tract infection. Diabetes was found not to be a risk factor for recurrent urinary tract infections in postmenopausal women.[16]
Bacteriological studies usually reveal the involvement of gram negative enteric organisms that commonly cause urinary tract infections, such as E. coli, the Klebsiella species, and the Proteus species.[17] Similarly, the predominant number of pathogens isolated in our study were gram negative bacilli rather than gram positive pathogens.
Among the patients infected with gram negative bacilli in our study, Escherichia coli was isolated from 71% of the subjects, Klebsiella spp. from 13.5%, Pseudomonas spp. from 9%, Enterobacter spp. and Citrobacter spp. in 2%, and nonfermenting gram negative bacilli and the Proteus spp in 1%.
In another study from India, it was found that E. coli was the most commounly grown organism (64.3%), followed by Staphylococcus aureus (21.4%), and Klebsiella pneumoniae (14.3%).[18] In a recent study, it was noted that increased adherence of E. coli with type 1 fimbriae to uroepithelial cells isolated from the urine of women with diabetes correlated positively with HbA1C. Poorly controlled patients had a higher adherence of E. coli.[19]
Urinary tract infections due to Enterococci are quite common, particularly in patients who have received antibiotic treatment or who have undergone instrumentation of the urinary tract.[20] It has been reported that the prevalence of enterococci as a cause of nosocomial UTI increased between 1975 and 1984.[21] Lloyds et al. have shown that Enterococcal species accounted for 35% of urinary tract isolates.[22] Our results showed that 59% of subjects had Enterococci spp. among gram positive pathogens.
Gram positive cocci play a lesser role in UTIs. However, Staphylococcs saprophyticus, a novobiocin-resistant, coagulase-negative species, accounts for 10–15% of acute symptomatic UTIs in young females.[23] Therapy with antibiotics directed at the offending organisms is important while the underlying diabetes is effectively managed.[24] Due to the frequent (symptomatic) upper tract involvement and the possibly serious complications, many experts recommend a 7–14 day oral antibacterial cystitis treatment in these patients with an antibacterial agent that achieves high concentrations both in the urine and in the urinary tract tissue. There is a great likelihood that UTIs are affected by antimicrobial resistance or atypical uropathogens due to which the risk of upper tract involvement is increased.[25]
The choice of antibiotic therapy should integrate the local sensitivity pattern of the infecting organisms. For seriously ill patients including patients infected with Pseudomonas, agents such as Imipenem, Ticarcillin-clavulanate, and Piperacillin-tazobactum may also be considered.
Treatment of asymptomatic bacteriuria in patients with diabetes is often recommended to prevent the risk of symptomatic UTIs.[26]
We also found that sulbactum/cefoperazone and pipercillin/tazobactum were highly sensitive to both gram positive cooci and gram negative bacilli.
Gram negative bacilli were found to be more sensitive than gram positive cocci to aminoglycosides such as netillin, amikacin, and tobramycin. Netilmycin is a derivative of Gentamycin that is less nephrotoxic and ototoxic. It is less active against Pseudomonas but it inhibits a number of strains of E. coli as well as Klebsiella resistant to tobramycin.[27]
Gram negative bacilli were found to be highly sensitive to ciprofloxacin (62%) than to ofloxacin (23%). Ciprofloxacin is thus clearly useful against polyresistant species such as Pseudomonas aeroginosa.[28] Gram positive cocci (63%) were found to be more sensitive to cefotaxime than gram negative bacilli (51%). Ceftriaxone, ceftizoxime, and cefotaxime have excellent activity against Streptococci.[17] More than two urinary tract infections per year should alert physicians to possible cystpathy and should elicit appropriate diagnostic procedures.[29]
One of the limitations of this study was that a control group was not included for comparison. In summary, the prevalence of lower UTI was high in women with type 2 diabetes than in men. UTI was found to be associated with age, duration of diabetes, and poor glycemic control. Escherichia coli was commonly isolated; the gram negative pathogens were highly sensitive to sulbactum / cefoperazone and piperacillin / tazobactum.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Bacteremia in diabetic patients: Comparison of incidence and mortality with non-diabetic patients. Diabetes Care. 1985;8:244-249.
- [Google Scholar]
- Acute Infections. In: Ahuja MMS, Tripathy BB, Sam Moses GP, Chandalia HB, Das AK, Rao PV, eds. RSSDI Text Book of Diabetes Mellitus. 2002. p. :471-8. Chap-34
- [Google Scholar]
- Epidemiology of urinary tract infections: Incidence, morbidity and economic costs, Dis. Mon. 2003;49:53-70.
- [Google Scholar]
- Emphysematous cystitis: A complication of urinary tract infection occurring predominantly in diabetic women. Ann Emerg Med. 1990;19:404-6.
- [Google Scholar]
- Asymptomatic bacteriuria may be considered a complication in women with diabetes. Diabetes Mellitus Women Asymptomatic Bacteriuria Utrecht-Study Group. Diab Care. 2000;23:744-9.
- [Google Scholar]
- Management of bacterial urinary tract infections in adult patients with diabetes mellitus. Drugs. 2002;62:1859-68.
- [Google Scholar]
- Urinary tract infection. Medicine, The Medicine publishing company Ltd; 2003. p. :85-90.
- The Kidney and Renal Tract. Diabetes: Clinical Mangement 8thedition, edited by 1990:286-7. Chap-25
- [Google Scholar]
- Definition, Diagnosis and Classification of diabetes mellitus and its complications. In: Part 1: Diagnosis and Classification of diabetes mellitus. Geneva: Department of non communicable Disease Surveillance. WHO; 1999.
- [Google Scholar]
- “Ü;ber die isolierte Färbung der Schizomyceten in Schnitt-und Trockenpräparaten” (in German) Fortschritte der Medizin. 1884;2:185-9.
- [Google Scholar]
- Antibiotic susceptibility testing by a standardised singles disk method. Am J Clin Pathol. 1966;45:493-6.
- [Google Scholar]
- Bailey and Scotts Diagnostic Microbiology (11th ed). 2002. p. :936-7.
- Bacterial urinary tract infections in diabetes. Infect Dis Clin North Am. 1995;9:25-51.
- [Google Scholar]
- Recurrent urinary tract infections in post menopausal women. Clin Infect Dis. 2000;30:152-6.
- [Google Scholar]
- Renal and perirenal infection: to he role of computerized tomography. J Urol. 1985;133:375-8.
- [Google Scholar]
- Prevalence of urinary tract infection and renal scars in patients with diabetes mellitus. Diab Res Clin Pract. 2001;53:181-6.
- [Google Scholar]
- Adherence of type 1-fimbriaeted E. coli to uro epithelial cells: Move in diabetic women than in control subjects. Diab care. 2002;25:1405-9.
- [Google Scholar]
- Wilson JD, ed. Streptococcal and enterococcal infections Harrison's principles of internal medicine 12 edition, edited by-. Vol Vol. 1. 1991. p. :901-9.
- Nosocomial urinary tract infection due to enterococcus. Arch Intern Med. 1986;146:1549-51.
- [Google Scholar]
- Risk factors for enterococal urinary tract infection and colonization in a rehabilitation facility. Am J Infect Control. 1998;26:35-9.
- [Google Scholar]
- Urinary tract infection and pyelonephritis. In: Braunwald E, ed. Harrison's Priniciples of Internal Medicine 15th edition, edited by-. Vol Vol. 2. 2001. p. :1620-26.
- [Google Scholar]
- Infections, Immunity, and Diabetes. In: International Text book of Diabetes Mellitus Vol Vol. 2. (3rd ed). 2004. p. :1729-39.
- [Google Scholar]
- Therapy and Prophylaxis of Bacterial infections. In: Wilson JD, ed. Harrison's Principles of Internal Medicine 12 edition, Nil - edited by-. Vol Vol. 478-92. 1991.
- [Google Scholar]
- A double blind study comparing two dosages of enoxacin for the treatment of uncomplicated urogenital gonorrhoea. J Antimicrob Chemother. 1984;14:91-4.
- [Google Scholar]
- Diabetic Automic Neuropathy. In: Diabetes mellitus Vol Vol. 46. (6th ed). 2003. p. :789-804.
- [Google Scholar]







