Translate this page into:
Repeat Transplantation with High-KDPI Kidneys: A Mate-Kidney Analysis
Corresponding author: Kalathil K Sureshkumar; Division of Nephrology and Hypertension, Medicine Institute, Allegheny General Hospital, Allegheny Health Network, Pittsburgh, PA, United States. E mail: kalathil.sureshkumar @ahn.org
-
Received: ,
Accepted: ,
How to cite this article: Sureshkumar KK, Chopra B, Josephson MA, Daloul R, McGill RL. Repeat Transplantation with High-KDPI Kidneys: A Mate-Kidney Analysis. Indian J Nephrol. doi: 10.25259/IJN_537_2024
Abstract
Background
Initial and subsequent kidney transplantation improves survival compared to continued dialysis. We compared initial and repeat deceased-donor kidney transplantation outcomes at different kidney donor profile index (KDPI) levels using an adjusted mate-kidney model.
Materials and Methods
Deceased donors whose kidneys were transplanted into adults were identified from the organ procurement and transplant network/united network for organ sharing database. Patients received peri-operative induction and maintenance tacrolimus/mycophenolate. Transplant pairs were stratified by KDPI: 0-20% (best), 21-85% (average), and 86-100% (marginal). Delayed graft function (DGF), graft failure, death, and graft failure with death as competing outcomes were compared between re-transplantation and first-time recipients.
Results
Transplantations from 52,218 donors to 104,436 recipients between 2000 and 2022 were included. In adjusted models, the DGF odds increased by 38% in repeat recipients (P<0.001). Hazard ratios (HR) for graft failure (HR:1.12, 95%confidence interval 1.07, 1.17), death (HR:1.14, 95%CI 1.09, 1.20), and graft failure with competing risk of death (HR:1.11, 95% CI 1.04, 1.18) were modest but significant (P<0.001). DGF and graft failure risk with competing death significantly increased with increasing KDPI.
Conclusion
Our study suggests that the long-term re-transplantation risks are modest. “Marginal kidneys” have the potential for re-transplantation in selected patients. Selective use of these kidneys would benefit patients undergoing re-transplantation.
Keywords
Graft outcomes
Mate-kidney model
Re-transplantation
Introduction
Compared to dialysis, kidney transplantation improves the longevity and quality of life.1-3 Better immunological risk assessment, immunosuppressive agents, and a broader therapeutic repertoire to manage immunological complications have caused improvement in long-term post-transplant outcomes.4 Repeat kidney transplant recipients are biologically more complex, with increased immunological risk, but benefit from the process.5,6 In 2020, the waiting list had 11.4% re-transplantation candidates.7
The kidney donor profile index (KDPI) is a metric for deceased donor kidney organ quality. KDPI scores, ranging from 0 to 100%, are calculated using donor specific factors from the kidney donor risk index developed by Rao et al.8 Lower scores indicate better quality. They are calculated using ten variables including donor age, height, weight, ethnicity, hypertension, diabetes, fatal cerebrovascular accident, serum creatinine, hepatitis C status, and whether donation occurred after circulatory death. Scores ranging from 86-100% are considered, “marginal” with non-utilization rates approaching 70%.7 These “marginal kidneys” generally provide inferior transplant outcomes compared to low KDPI kidneys.7 However, transplantation with these, as opposed to waiting, confers survival advantages.9 It is intuitive that ‘marginal kidneys’ would give better outcomes when transplanted into first-time recipients as compared to those with failed transplants.
Donor organ quality is a major determinant in allograft function and long-term survival.10-12 To minimize their impact, investigators usually try to adjust for different donor variables in analysis. Hemodynamic instability during organ procurement, vasoactive medications, and donor kidney histology are indeterminable factors affecting transplant outcomes.13,14 Confounding effects of donor variables could be reduced by comparing recipients’ transplant outcomes of mate-kidneys from a common deceased donor.
The study uses a mate-kidney model to quantify the differences between outcomes of initial and repeat deceased-donor kidney transplantations at different donor KDPI levels. We attempted to evaluate the advisability of using high KDPI kidneys for re-transplantation.
Materials and Methods
The study protocol was approved by the Institutional Review Boards of the Allegheny Health Network and the University of Chicago and performed adhering to the Declaration of Helsinki, with a waiver of informed consent for using de-identified data. Deceased donors whose kidneys were transplanted into adults between January 2000 and December 2022 were identified from the Organ Procurement and Transplant Network (OPTN)/United Network for Organ Sharing database, the time frame chosen to reflect contemporary immunosuppression and management. We restricted our analysis to patients with KDPI and delayed graft function (DGF) data who received perioperative antibody induction followed by maintenance immunosuppressive therapy with tacrolimus and mycophenolic acid. Transplant pairs were classified as concordant (first-time or repeat recipients) or discordant (one first-time and one repeat recipient) and stratified by KDPI: 0-20% (best kidneys); 21-85% (average kidneys); 86-100% (“marginal kidneys”). Among re-transplant recipients, the original transplant could be from a living or deceased donor. Length of stay, DGF (the need for dialysis during first week post-transplant), graft failure, and patient death were assessed. Graft failure with competing death risk was also calculated, in lieu of a death-censored graft failure.
Statistical analysis
Baseline characteristics were compared between initial and repeat kidney recipients, using t-tests, Mann-Whitney U tests, and chi-squared tests, as appropriate. Baseline characteristics in discordant recipients were compared using paired-t tests, Wilcoxon signed-rank tests, McNemar’s and Bowker’s tests, as appropriate. Time to event outcomes of graft failure and death, with death as a competing risk, were evaluated with marginal models, paired by donor ID with robust sandwich estimators and sub distributions hazard model using Fine and Grey methods, respectively. Regression approaches were optimized for each outcome. Conventional survival analysis estimates the probability of the event of interest, given that no other event has occurred. When modeling the hazard of mortality, censoring for graft failure would be inappropriate. Patients lost to follow-ups are properly censored, as their ultimate outcomes are unknown. When modeling graft failure, a patient who dies with a functioning graft has a known final graft outcome and is no longer at risk for graft failure. Therefore, death is a true competing outcome for graft failure, and censoring at the time of death would inflate the probability of graft failure. Competing risks methodology calculates the cumulative graft failure incidence, which is a more accurate estimate of risk.15 Times to event were calculated from the date of kidney transplantation until graft failure, return to dialysis, death, or December 31, 2022. DGF was evaluated with conditional logistic regression. Initial transplant recipients were the reference group, hazard ratios (HR) and odds ratios (OR) were calculated for re-transplantation. Interaction variables between KDPI category and initial vs. repeat transplantation were constructed, allowing calculation of KDPI-specific custom HRs for re-transplantation at each level for each survival outcome. KDPI-specific odds ratios for comparing DGF between re-transplantation and first-time recipients were obtained by stratified conditional logistic models. For sensitivity analysis, all models were also performed on discordant matched pairs (with one first-time and one repeat recipient) exclusively. All models were adjusted for recipient age, sex, race, body mass index (BMI), insurance status, panel reactive antibody (PRA) titer (a measure of recipient sensitization), primary kidney diagnosis, diabetes, peripheral vascular disease (PVD), recipient viral sero-status for cytomegalovirus (CMV), hepatitis B and C viruses (HBV, HCV), distance from transplant center, right/left donor kidney, pump perfusion, cold ischemia time, ABO matching, human leukocyte antigen (HLA), and DR mismatches, lymphocyte depleting induction and steroid maintenance. Missing data were handled using the multiple imputation Markov Chain Monte Carlo (MCMC) method, generating ten data sets. P-values <0.05 were considered significant. Analysis was done using SAS 9.4 (SAS, Cary, NC).
Results
The median study follow-up was 49 months (interquartile range = 22-92). The flow chart for inclusion is shown in Figure 1. Inclusion criteria were met by 104,436 recipients with 52,218 donors, including 18,786 discordant recipients with 9,393 donors (18.0%). Distribution of KDPI categories was as follows: best kidneys (KDPI 0-20%) =23.1%; average kidneys (KDPI 21-85%) =71.2%; “marginal kidneys” (KDPI 86-100%) =5.7%. Donor variables are shown in Supplementary Table 1.
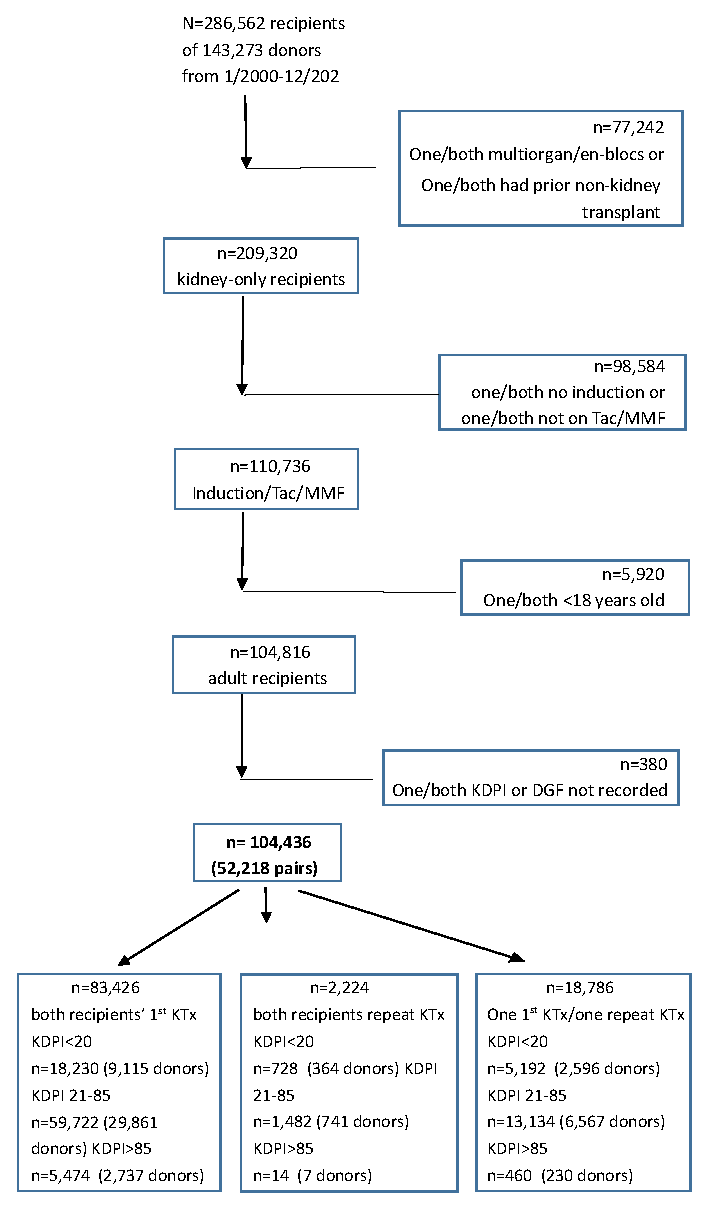
- Flow chart for paired patient selection. DGF: Delayed graft function, KDPI: Kidney donor profile index; KTx: Kidney transplantation; MMF: Mycophenolate mofetil.
Characteristics of the study groups stratified by first-time versus repeat recipients are shown in Table 1. Repeat recipients were younger, more frequently CMV and HCV seropositive, and more likely to be female and white. They had lower BMI and less frequent PVD but were less likely to have listed primary kidney disease. Fewer than one-fourth of patients in either group had private insurance. Repeat recipients had fewer HLA and DR mismatches, higher PRA titers, and were more likely to receive depleting induction and maintenance corticosteroids. Repeat recipients were more likely to receive left kidneys and less likely to receive pump-perfused kidneys, with longer traveling and cold ischemia times. First-time recipients were more likely to receive marginal KDPI kidneys than repeat ones (2.1% vs. 6.2%, P<0.001). Similar findings were seen among the discordant mate-kidney pairs subset [Table 2].
| All patients | First transplant | Repeat transplant | P | |
|---|---|---|---|---|
| Number of participants | 104,436 | 92,819 | 11,617 | |
| Age (years) | 53 ± 13 | 54 ± 13 | 47 ± 13 | <0.001 |
| Sex (% female) | 39.1 | 38.9 | 40.4 | 0.002 |
| Race, % | ||||
|
White African American Hispanic Asian Others |
38.2 34.1 18.4 7.2 2.1 |
37.0 34.6 18.8 7.5 2.1 |
47.8 29.6 15.6 5.2 1.8 |
<0.001 |
| Primary disease, % | ||||
|
Diabetes Hypertension Primary GN Secondary GN Cystic disease Other |
28.9 25.0 13.9 4.6 7.8 19.8 |
32.0 27.0 14.1 4.9 8.4 13.6 |
3.9 9.4 12.0 2.8 3.0 68.9 |
<0.001 |
| Other diabetes, % | 7.5 | 6.7 | 14.4 | <0.001 |
| Peripheral vascular disease, % | 9.1 | 9.4 | 6.2 | <0.001 |
| Body mass index | 28.3 (5.4) | 28.5 (5.4) | 26.9 (5.3) | <0.001 |
| Private insurance, % | 23.5 | 23.6 | 22.8 | 0.06 |
| CMV matching, % | ||||
|
D+/R- D+/R+ or D-/R+ D-/R- Unknown |
17.5 69.3 12.1 1.1 |
17.8 68.9 12.2 1.1 |
14.8 72.9 10.9 1.4 |
<0.001 |
| HBV+, % | 8.8 | 8.9 | 7.4 | <0.001 |
| HCV+ | 4.2 | 4.1 | 4.8 | 0.003 |
| Panel reactive antibody %, median (IQR) | 0 (0,45) | 0 (0,24) | 89 (49, 99) | <0.001 |
| ABO match, % | ||||
|
Identical Compatible Incompatible |
95.1 3.5 1.4 |
95.4 3.2 1.4 |
93.4 6.0 0.6 |
<0.001 |
| HLA mismatches, % | ||||
|
0 1 2 3 4 5 6 |
6.0 1.6 4.5 13.6 27.3 32.0 15.0 |
5.1 1.3 4.0 13.1 27.5 33.2 15.8 |
12.9 4.6 8.6 17.9 25.8 22.0 8.2 |
<0.001 |
| DR mismatches, % | ||||
|
0 1 2 |
17.7 47.2 35.1 |
16.0 47.7 36.3 |
31.6 42.9 25.5 |
<0.001 |
| Cold ischemia hours | 18 ± 9 | 18 ± 9 | 19 ± 8 | 0.008 |
| Miles from center, median (IQR) | 63 (8,188) | 60 (8, 175) | 102 (13, 412) | <0.001 |
| Left kidney, % | 50.0 | 49.6 | 53.5 | <0.001 |
| On-pump, % | 50.0 | 50.7 | 43.8 | <0.001 |
| Lymphocyte depleting induction | 80.8 | 79.5 | 90.9 | <0.001 |
| Maintenance steroids, % | 73.4 | 72.2 | 82.6 | <0.001 |
| KDPI | ||||
|
≤ 20 21-85 >85 |
23.1 71.8 5.7 |
22.4 71.4 6.2 |
28.6 69.3 2.1 |
<0.001 |
GN: Glomerulonephritis, D+/D-: Donor-positive/negative; R+/R-: Recipient-positive/negative, HBV: Hepatitis B, HCV: Hepatitis C, IQR: Interquartile range, HLA/DR: Human leukocyte antigens/DR isotypes, KDPI: Kidney donor profile index, CMV: Cytomegalo virus.
| All patients | First transplant | Repeat transplant | P | |
|---|---|---|---|---|
| Number of participants | 18,786 | 9,393 | 9,393 | |
| Age (years) | 50 ± 13 | 53 ± 13 | 47 ± 13 | <0.001 |
| Sex (% female) | 42.5 | 45.0 | 39.9 | <0.001 |
| Race, % | ||||
|
White African American Hispanic Asian Others |
43.4 31.9 16.8 6.0 1.9 |
38.8 34.4 18.0 6.8 2.0 |
47.9 29.4 15.6 5.3 1.8 |
<0.001 |
| Primary disease, % | ||||
|
Diabetes Hypertension Primary GN Secondary GN Cystic disease Other |
16.6 17.9 13.2 4.2 5.9 42.2 |
29.3 26.5 14.6 5.7 8.7 15.3 |
4.0 9.3 11.8 2.8 3.1 69.1 |
<0.001 |
| Other diabetes, % | 10.5 | 6.5 | 14.5 | <0.001 |
| Peripheral vascular disease, % | 7.7 | 9.1 | 6.3 | <0.001 |
| Body mass index | 27.7±5.5 | 28.5±5.5 | 26.9±5.3 | <0.001 |
| Private insurance, % | 23.5 | 24.1 | 22.8 | 0.03 |
| CMV matching, % | ||||
|
D+/R- D+/R+ or D-/R+ D-/R- Unknown |
16.1 70.6 11.9 1.4 |
17.2 68.4 13.0 1.4 |
14.9 72.8 10.8 1.5 |
<0.001 |
| HBV+, % | 8.0 | 8.5 | 7.5 | 0.002 |
| HCV+ | 4.5 | 4.0 | 5.0 | <0.001 |
| Panel reactive antibody %, median (IQR) | 50 (0, 94) | 0 (0, 60) | 86 (38, 98) | <0.001 |
| ABO match, % | ||||
|
Identical Compatible Incompatible |
94.1 5.0 0.9 |
94.6 4.3 1.1 |
93.6 5.7 0.7 |
<0.001 |
| HLA mismatches, % | ||||
|
0 1 2 3 4 5 6 |
11.0 3.2 6.6 16.4 26.4 25.6 10.8 |
10.1 2.3 5.1 15.2 26.4 28.2 12.7 |
11.9 4.0 8.0 17.7 26.5 23.0 8.9 |
<0.001 |
| DR mismatches, % | ||||
|
0 1 2 |
23.8 44.2 29.0 |
24.0 45.0 31.0 |
29.6 43.3 27.1 |
<0.001 |
| Cold ischemia hours | 18 ± 9 | 18 ± 9 | 18 ± 9 | 0.4 |
| Miles from center, median (IQR) | 81 (10, 263) | 67 (9, 207) | 92 (12, 352) | <0.001 |
| Left kidney, % | 50.0 | 45.6 | 54.4 | <0.001 |
| On-pump, % | 44.8 | 45.2 | 44.4 | 0.06 |
| Lymphocyte depleting induction, % | 85.3 | 80.0 | 90.5 | <0.001 |
| Maintenance steroids, % | 77.6 | 72.7 | 82.5 | <0.001 |
| KDPI | ||||
|
≤20 21-85 >85 |
27.6 69.9 2.5 |
GN: Glomerulonephritis, D+/D-: Donor-positive/negative, R+/R-: Recipient-positive/negative, HBV: Hepatitis B, HCV: Hepatitis C, IQR: Interquartile range, HLA: Human leukocyte antigens, KDPI: Kidney donor profile index, CMV: Cytomegalo virus.
Outcomes
Hospital length of stays for repeat and first-time transplant patients were 6.95± 9.09 vs. 6.57± 9.57 days, respectovely (P<0.001). Outcomes from unadjusted and adjusted models have been shown in Table 3. Odds of DGF in adjusted models was 1.38 (95%CI=1.26, 1.53, P<0.001) for repeat vs. first-time recipients. Adjusted HRs for graft failure (1.12, 95%CI=1.07,1.17), death (1.14, 95%CI=1.09,1.20), and graft failure with competing death (1.11, 95%CI=1.04-1.18) were significantly higher in repeat transplant recipients. Unadjusted graft failure and death were significantly lower for repeat recipients, suggesting rigorous patient selection for repeat transplantation.
| Unadjusted | Adjusted | |||
|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | |
| Graft failure | 0.94 (0.91, 0.98) | <0.001 | 1.12 (1.07, 1.17) | <0.001 |
| Death | 0.79 (0.75, 0.82) | <0.001 | 1.14 (1.09, 1.20) | <0.001 |
| Graft failure, competing death | 1.11 (1.06, 1.17) | <0.001 | 1.11 (1.04, 1.18) | <0.001 |
| OR (95% CI) | P | OR (95% CI) | P | |
| Delayed graft function | 1.23 (1.14, 1.32) | <0.001 | 1.38 (1.26, 1.53) | <0.001 |
HR: Hazard ratio, CI: Confidence interval, OR: Odds ratio.
Outcomes conditioned on KDPI categories are shown in Table 4. Long-term outcomes for both types of recipients of optimal kidneys with KDPI ≤ 20% were uniformly good; however, repeat transplantation was associated with higher DGF (OR: 1.49, 95%CI=1.20, 1.85, P<0.001). For average allograft recipients (KDPI 21-85%), re-transplantation was associated with higher risks of death (HR: 1.15, 95% CI = 1.08, 1.23, p<0.001), graft failure (HR: 1.16, 95% CI = 1.10, 1.23, p<0.001), graft failure with competing death (HR: 1.12, 95% CI = 1.05, 1.20, p 0.001), and DGF (OR: 1.36, 95% CI =1.21, 1.52, p <0.001). Among marginal kidney recipients, re-transplantation was associated with higher risks of death (HR: 1.16, 95% CI = 0.96, 1.48, p 0.2), graft failure (HR: 1.08, 95% CI = 0.99, 1.18, p 0.08) and graft failure with competing death (HR: 1.27, 95% CI = 1.00, 1.61, p 0.05) but these associations were not statistically significant, reflecting wider confidence intervals due to smaller number of patients. Re-transplantation was associated with a significant risk of DGF in marginal kidney recipients (OR: 1.95, 95%CI=1.12, 3.40).
| KDPI category | Best (≤ 20) | Average (21-85) | Marginal (> 85) | |||
|---|---|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | HR (95% CI) | P | |
| Death | 1.00 (0.89, 1.11) | 0.9 | 1.15 (1.08, 1.23) | <0.001 | 1.16 (0.92, 1.48) | 0.2 |
| Graft failure | 1.08 (0.99, 1.18) | 0.08 | 1.16 (1.10, 1.23) | <0.001 | 1.13 (0.92, 1.38) | 0.2 |
| Graft failure, Competing death | 1.05 (0.94, 1.16) | 0.4 | 1.12 (1.05, 1.20) | 0.001 | 1.27 (1.00, 1.61) | 0.05 |
| OR (95% CI) | P | OR (95% CI) | P | OR (95% CI) | P | |
| Delayed graft function | 1.49 (1.20, 1.85) | <0.001 | 1.36 (1.21, 1.52) | <0.001 | 1.95 (1.12, 3.40) | 0.02 |
HR: Hazard ratio, CI: Confidence interval, OR: Odds ratio, KDPI: Kidney donor profile index. Adjusted for: recipient age, sex, body mass index, insurance, panel reactive antibody, primary kidney diagnosis, diabetes, peripheral vascular disease, distance from center, right/left donor kidney, pump perfusion, cold ischemia time, ABO matching, HLA/DR mismatching, lymphocyte depletion, glucocorticoid maintenance and viral serologies for cytomegalovirus and hepatitis B and C.
Sensitivity analysis
In sensitivity analyses, effect sizes were equivalent to primary analyses [Table 5]. However, the smaller analytical sample size resulted in broader confidence intervals, with insufficient statistical power for the outcome of graft failure with competing death (HR: 1.08 (95% CI=0.98, 1.19, P=0.1).
| Unadjusted | Adjusted | |||
|---|---|---|---|---|
| HR (95% CI) | P | HR (95% CI) | P | |
| Graft failure | 1.00 (0.95, 1.05) | 0.9 | 1.11 (1.03, 1.21) | 0.009 |
| Death | 0.84 (0.80, 0.89) | <0.001 | 1.16 (1.06, 1.27) | <0.001 |
| Graft failure, death competing | 1.16 (1.08, 1.24) | <0.001 | 1.08 (0.98, 1.19) | 0.1 |
| OR (95% CI) | P | OR (95% CI) | P | |
| Delayed graft function | 1.23 (1.14, 1.32) | <0.001 | 1.31 (1.13, 1.52) | <0.001 |
HR: Hazard ratio, CI: Confidence interval, OR: Odds ratio
Discussion
We used a mate-kidney model to quantify the effect of interactions between allograft quality and re-transplantation on allograft outcomes. Re-transplantation was associated with significant increases in adverse long-term patient and graft outcomes and a substantial increase in DGF. Using the mate-kidney model, we separated the effects of allograft quality from those of complexities of repeat transplantation (increased recipient age and comorbid disease burden, surgical risks, additional exposure to lymphocyte depleting agents, and more DGF).
Decreased patient survival in repeat recipients has been reported in previous studies regardless of KDPI.16 Repeat kidney transplantation confers significant survival benefits over dialysis despite inferior transplant outcomes.6,16-19 The 10-15% increase in adverse long-term outcomes observed with re-transplants should be viewed in light of its survival benefits over dialysis.5,6,16-19 Graft and patient outcomes were similar for repeat and first-time recipients in the best KDPI group despite increased risk for DGF with re-transplantation. The risks associated with re-transplantation were most clearly demonstrated in the large pool of recipients receiving kidneys with KDPI 21-85%. These differences were more difficult to demonstrate among marginal kidney recipients due to the lack of their use in repeat transplants. Only 2.1% of the re-transplants were with kidneys with KDPI 86-100%.
This analysis is clinically relevant as “marginal kidneys” have high non-utilization rates7, and patients wait-listed for re-transplantation generally have increased risks for delisting and death due to larger burdens of comorbidities. The benefits are more pronounced with preemptive re-transplantation20,21 and diminish as the patient receives dialysis while waiting on the list.18,19,22 Preemptive kidney transplantation with KDPI ≥85% has yielded outcomes like non-preemptive transplantation with lower KDPI kidneys.23,24 Accepting marginal kidneys more liberally for repeat recipients could potentially decrease waiting times and increase access to re-transplantation.
Recipient characteristics impact the potential benefit of receiving a marginal kidney. Previous studies have suggested patient groups like younger and non-diabetic patients, expected to have longer survival with or without transplants, might not benefit much from receiving marginal kidneys.25-27 However, patients >60 years,28 with high estimated post-transplant survival score (EPTS >80%) and shorter recipient survival expectation, might benefit lifelong from receiving a marginal kidney, compared to dialysis.29 Our results, in which the unadjusted death and graft failure were superior among repeat recipients, suggest that current practices may provide a repeat transplantation recipient pool that is more highly selected than initial recipients. Expanding the use of marginal kidney allografts would require thoughtful consideration by the transplant community. The use of high-KDPI kidneys for patients with a failed kidney transplant merits further risk-benefit analysis.
Patients with failed kidney transplants are at increased risk due to higher rates of comorbidities and sensitization status. Intuitively, some physicians might hesitate to accept high-KDPI kidneys to avoid simultaneous disadvantages in the allograft and recipient. A registry study utilizing data from the Scientific Registry of Transplant Recipients suggested that using expanded criteria donor (ECD) kidneys was not associated with survival benefit when compared to remaining on the waiting list.5 This analysis of an earlier patient cohort (transplantation between 1995-2004) had older ECD kidney recipients with a higher diabetes prevalence of diabetes. Our study used a large national United States registry that reflects contemporary clinical practice and examined the interaction between different KDPI and re-transplant levels.
The study’s strengths are the large patient population and the use of a mate-kidney model adjusted for many relevant variables. Nonetheless, certain limitations should be acknowledged. Our study design is retrospective, providing associations rather than causation. Selection bias was likely among re-transplantation candidates. Data regarding donor specific antibodies, etiologies/severity of rejection episodes, or the doses of induction and immunosuppressive medications was unavailable. Our mate-kidney model could not account for important variables such as warm ischemia time or the surgical techniques and experience of individual transplant centers. Given the mate-kidney model used in the analysis, outcomes of marginal kidney transplants in which the mate kidneys were not transplanted were not examined.
Our results suggest that “marginal kidneys” could be retransplanted in selected patients. Although re-transplantation is associated with inferior outcomes, effect sizes are small and compare favorably to the risks inherent in remaining on dialysis. Recipient characteristics, such as age, cardiovascular status, and diabetes, should be considered when contemplating a “marginal kidney” for a recipient with history of a previous kidney transplant.
Acknowledgments
Presented in part at ASN Kidney Week November 2022, Orlando, FL. The data reported here have been supplied by the United Network for Organ Sharing as the contractor for the Organ Procurement and Transplant Network. The interpretation and reporting of these data are the responsibility of the authors and in no way should be seen as an official policy of or interpretation by OPTN or the US Government.
Conflicts of interest
There are no conflicts of interest.
References
- Impact of cadaveric renal transplantation on survival in patients listed for transplantation. J Am Soc Nephrol. 2005;16:1859-65.
- [CrossRef] [PubMed] [Google Scholar]
- Health-related quality of life after solid organ transplantation: A prospective, multiorgan cohort study. Transplantation. 2013;96:316-23.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of transplantation on quality of life in patients with diabetes and renal dysfunction. Transplantation. 2000;70:1736-46.
- [CrossRef] [PubMed] [Google Scholar]
- Long-term survival after kidney transplantation. N Engl J Med. 2021;385:729-43.
- [CrossRef] [PubMed] [Google Scholar]
- Mortality experience in recipients undergoing repeat transplantation with expanded criteria donor and non-ECD deceased-donor kidneys. Am J Transplant. 2007;7:1140-7.
- [CrossRef] [PubMed] [Google Scholar]
- Access to kidney transplantation after a failed first kidney transplant and associations with patient and allograft survival: An analysis of national data to inform allocation policy. Clin J Am Soc Nephrol. 2019;14:1228-37.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- OPTN/SRTR 2020 Annual Data Report, 10 March 2022.
- A comprehensive risk quantification score for deceased donor kidneys: The kidney donor risk index. Transplantation. 2009;88:231-6.
- [CrossRef] [PubMed] [Google Scholar]
- Survival in recipients of marginal cadaveric donor kidneys compared with other recipients and wait-listed transplant candidates. J Am Soc Nephrol. 2001;12:589-97.
- [CrossRef] [PubMed] [Google Scholar]
- Preoperative assessment of the deceased-donor kidney: From macroscopic appearance to molecular biomarkers. Transplantation. 2014;97:797-807.
- [CrossRef] [PubMed] [Google Scholar]
- Validation of the Maryland aggregate pathology index (MAPI), a pre-implantation scoring system that predicts graft outcome. Clin Transplant. 2014;28:897-905.
- [CrossRef] [PubMed] [Google Scholar]
- Histopathologic evaluation of pretransplantation biopsy as a factor influencing graft function after kidney transplantation in 3-year observation. Transplant Proc. 2010;42:3375-81.
- [CrossRef] [PubMed] [Google Scholar]
- A simple clinico-histopathological composite scoring system is highly predictive of graft outcomes in marginal donors. Am J Transplant. 2008;8:2325-34.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluation of pre-implantation kidney biopsies: Comparison of Banff criteria to a morphometric approach. Kidney Int. 2005;67:1595-600.
- [CrossRef] [PubMed] [Google Scholar]
- Competing risk analysis in renal allograft survival: A new perspective to an old problem. Transplantation. 2021;105:668-76.
- [CrossRef] [PubMed] [Google Scholar]
- Re-transplants compared to primary kidney transplants recipients: A mate kidney paired analysis of the OPTN/UNOS database. Clin Transplant. 2016;30:566-78.
- [CrossRef] [PubMed] [Google Scholar]
- Prognosis after primary renal transplant failure and the beneficial effects of repeat transplantation: Multivariate analyses from the United States renal data system. Transplantation. 1998;66:1651-9.
- [CrossRef] [PubMed] [Google Scholar]
- Waiting time for second kidney transplantation and mortality. Clin J Am Soc Nephrol. 2022;17:90-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Dialysis vintage and outcomes after kidney transplantation: A retrospective cohort study. Clin J Am Soc Nephrol. 2017;12:122-30.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Preemptive second kidney transplantation is associated with better graft survival compared with non-preemptive second transplantation: A multicenter French 2000-2014 cohort study. Transpl Int. 2018;31:408-23.
- [CrossRef] [PubMed] [Google Scholar]
- Risks and benefits of preemptive second kidney transplantation. Transplantation. 2013;95:705-10.
- [CrossRef] [PubMed] [Google Scholar]
- Waiting time between failure of first graft and second kidney transplant and graft and patient survival. Transplantation. 2016;100:1767-75.
- [CrossRef] [PubMed] [Google Scholar]
- Kidney transplantation in older recipients: Preemptive high KDPI kidney vs lower KDPI kidney after varying dialysis vintage. World J Transplant. 2018;8:102-9.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- The Benefits of preemptive transplantation using high-kidney donor profile index kidneys. Clin J Am Soc Nephrol. 2023;18:634-43.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Deceased-donor characteristics and the survival benefit of kidney transplantation. JAMA. 2005;294:2726-33.
- [CrossRef] [PubMed] [Google Scholar]
- High mortality in diabetic recipients of high KDPI deceased donor kidneys. Clin Transplant. 2016;30:940-5.
- [CrossRef] [PubMed] [Google Scholar]
- Which renal transplant candidates should accept marginal kidneys in exchange for a shorter waiting time on dialysis? Clin J Am Soc Nephrol. 2006;1:532-8.
- [CrossRef] [PubMed] [Google Scholar]
- Survival benefit in older patients associated with earlier transplant with high KDPI kidneys. Transplantation. 2017;101:867-72.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Who can tolerate a marginal kidney? Predicting survival after deceased donor kidney transplant by donor-recipient combination. Am J Transplant. 2019;19:425-33.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]








