Translate this page into:
Survival of Tunneled Double Lumen-Cuffed Catheters in Children on Maintenance Hemodialysis – A Retrospective Cohort Study
Address for correspondence: Dr. Richa Prakash, 1A/1503, Green Acres, Lokhandwala Complex, Andheri West, Mumbai, Maharashtra, India. E-mail: rch.prakash@gmail.com
-
Received: ,
Accepted: ,
This article was originally published by Wolters Kluwer - Medknow and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Introduction:
Survival of tunneled cuffed catheters (TCC), used widely in children, is complicated by infections and catheter dysfunction. In resource limited settings, where risk of complications could be higher and waiting period for transplantation longer, catheter survival determines patient survival. This study was conducted to determine infection free catheter survival rates, incidence of catheter failure and associated risk factors.
Methods:
Children <18 years of age receiving maintenance hemodialysis through TCC at nephrology division of a pediatric hospital, over a period of 6 years. Data was collected with consecutive selection by a complete enumeration technique from pre-collected data sheets in the records. Exposure detected were catheter infections, thrombosis, and mechanical complications.
Results:
Forty-five TCCs in 36 children studied for 12,590 catheter days showed catheter failure in 36%, due to catheter related infections in 75% and mechanical complications in 25%. The incidence of complications per 1000 catheter days was 1.19 infection, 1.03 thrombus, and 0.39 mechanical. Catheter-related blood stream infection (CRBSI) (15/36) was associated with thrombus in nine and led to mortality in three. The mean infection free catheter survival was 449 ± 42 days for cohort with 388 ± 38 days in Group A (premature catheter removal) and 593 ± 43 days in Group B (elective removal) (P = 0.03). Catheterization duration of 267 days predicted CRBSI (sensitivity 93%, specificity 66.7%) with area under the curve of 0.808.
Conclusions:
Median infection free catheter survival was 449 days with catheter failure in 36%. CRBSI was the main cause of failure. Duration of catheterization greater than 267 days was a predictor of CRBSI.
Keywords
Catheter failure
catheter-related blood stream infection
catheter-related complications
cuffed haemodialysis catheters
thrombus
Introduction
Despite strong recommendations of fistula first, tunneled cuffed catheters (TCC) remains the commonest vascular access used globally for maintenance hemodialysis (HD) in children.[1] Boryzch-Duzalka et al.[2] consolidated data from 27 European countries performing HD in children and found that despite a gradual increase in the creation of arterio-venous fistula (AVF), 90% of children below 10 years of age still continued to have TCC as access for HD. In the US, almost 80% of children continue to be dialyzed through a TCC.[34] Catheter-related blood stream infections (CRBSIs) have been an important cause of premature catheter removal, increased morbidity, hospitalization, and death rates.[5678] Catheter malfunction due to thrombus, malposition, or kink are the other major reasons for catheter failure.[9101112]
Creation of AVF in pediatric population has a major challenge in the narrow caliber vessels which requires expert personnel. Owing to this, the use of TCCs for HD is as high as 87–89% in few developing nations.[2] Reports from single center studies show a heterogeneous outcome with some reporting catheter survival rates in middle and low-income countries to be comparable to those in high-income countries, while others have reported higher rates of CRBSI or shorter catheter survival rates.[91314]
In India, there has been a slow but steady increase in the number of centers offering maintenance HD for children in the last 10–12 years. Outcome studies are scarce and have shown a high mortality and dropout rates.[1516] Despite the wide use of tunneled catheters, the incidence of catheter-related complications and the catheter survival rates are not known.
This study was undertaken to determine the infection free survival rate of TCCs, the incidence and causes of catheter failure, and the associated risk factors in children undergoing maintenance HD through tunneled catheters at our pediatric dialysis center in a public hospital serving children from a low socioeconomic background.
Methodology
This was a retrospective cohort study conducted at the pediatric nephrology division of a tertiary care children’s hospital in Mumbai. The study was conducted over a 6-year period from September 1, 2012 to September 30, 2018. Subjects included all children who had a TCC inserted at our hospital for maintenance HD and received treatment for >3 months. Patients were selected consecutively by the complete enumeration sampling technique from the dialysis records. Children who opted for dialysis treatment at a different center within 3 months of catheter insertion were excluded from the study [Figure 1].
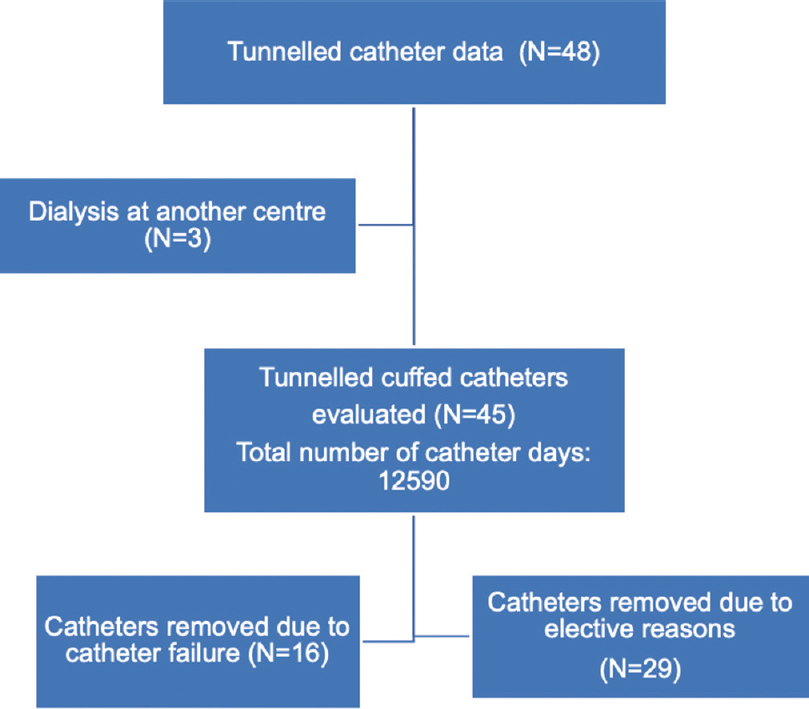
- Flow chart explaining the data collection
Data was collected from the medical records, from the time of catheter insertion until the time of removal, or in case of persistent patency, till last follow up. The following details were noted: 1) patient characteristics – age, gender, primary renal diagnosis, indication for dialysis, 2) catheter details – catheter size, laterality, catheter type, 3) incidence of catheter-related complications including infection, thrombus, and mechanical issues, and 4) survival of catheter until removal and infection free survival of the catheters. Patients were categorized into two groups. The first group (A) underwent “premature catheter removal” due to the various catheter-related complications and the second group (B) consisting of those who were without premature catheter removal, that is, catheters were removed when it was no longer needed or remained functional till the last follow up. The study was approved by the institutional ethics committee of the hospital.
Inclusion criteria
All children <18 years of age who underwent maintenance hemodialysis for end stage kidney disease (ESRD) via TCCs inserted in hospital were enrolled.
Exclusion criteria
The following patients were excluded. 1) Patients in whom HD catheter placement had been done at an outside hospital, 2) patients who were taking dialysis at a different center, and 3) patients with incomplete dialysis records, or lost to follow up within 3 months of catheter insertion.
Definitions used in the study
Catheter survival was the number of days of the catheter from the date of catheter insertion to the date of catheter exchange, removal or last follow up in case of patency. Infection free catheter survival was calculated as the number of days of indwelling catheter from the date of catheter placement to the first episode of catheter related infection. Catheter failure was defined as premature removal of TCC due to any catheter-related complication. CRBSI was diagnosed in patients with clinical manifestations of infection such as fever without focus or chills during dialysis along with positive cultures obtained from both peripheral blood and catheter hub blood, with growth of the same microorganism, identical in species and antibiogram from both sites (both collected simultaneously) and with the hub blood culture yielding positive results 2 h earlier than peripheral culture.[1718]
Catheter dysfunction was diagnosed when at least one of the following criteria were met: 1) inability to initiate dialysis due to inadequate blood flow or 2) inability to reach the blood flows which had been previously achieved.[1920]
Type of catheters
Permanent indwelling catheters inserted were either polycarbonate-urethane Bard hemosplit Equistream (CR Bard, Arizona, USA) or silicone Mahurkar (Covidien, USA) or silicone Medcomp (Harylsville, PA, USA), according to availability with the size decided according to the patient’s age. The choice of catheters was based on the catheters readily available at our center.
Routine catheter care was performed at the end of each dialysis session. The exit site was cleaned with a solution of 2.5% chlorhexidine in alcohol followed by a sterile transparent dressing of the exit site that was changed weekly unless indicated earlier. The catheter was locked with heparin in a concentration of 5000 units per milliliter.
Management of complications
CRBSI: Empirical antibiotics administered were either a third-generation cephalosporin or a beta-lactam inhibitor combination, due to the high prevalence of gram-negative organisms in our patient population. After identification of the organism and obtaining the final sensitivity reports, treatment was modified based on the sensitivity report.
Catheter was removed in all cases where pseudomonas or fungus was isolated, in patients who were hemodynamically unstable, in the presence of signs of sepsis despite 72 h of appropriate systemic antibiotics or in the presence of metastatic infections. If at 72 h, the patient was afebrile and hemodynamically stable, TCC was retained with an antibiotic lock consisting of gentamycin plus heparin, as majority of the isolated organisms isolated were gram negative with sensitivity to gentamicin according to antibiogram.[21] Antimicrobials were continued for 2–6 weeks depending on the organism isolated.[20]
Thrombus: Doppler studies were performed at time of catheter dysfunction or during an ongoing CRBSI episode. Occlusive intramural thrombi were treated initially with urokinase locking solutions for 1-h dwells. In unresolved cases, thrombolysis was attempted with recombinant-urokinase infusions, in the absence of active infection.[202223]
Mechanical complications: Kink and malposition were initially managed with guidewire manipulations, failing which the catheter was removed. All cracked catheters were immediately replaced with another catheter at the same or different site.[20]
Statistical analysis
The continuous variables were expressed as mean ± SD and the categorical variables as frequencies or percentage. Fisher’s exact test or Chi-square test was used for comparing categorical variables in the two groups and student’s t-test for continuous variables. Survival curves for primary catheter patency and infection free survival were computed using Kaplan and Meier methodology and comparisons analyzed using the Log Rank test. The binary regression model was used to identify significant variables affecting the incidence of infection and the goodness-of-fit model tested using the Hosmer–Lemeshow test. The receiver operating characteristic (ROC) curve was constructed to determine the predictive strength of duration of catheterization for CRBSI. All statistical tests were done on SPSS v 25.0 software, and P value <0.05 was considered significant.
Results
Forty-five TCCs inserted were studied for 12,590 catheter days. Of the 48 TCCs inserted during the study period, three continued at a different dialysis center within the first 3 months and were excluded from the study [Figure 1]. Apart from these patients, around 18 patients with ESRD were initiated on CAPD but none received a preemptive fistula formation, during the same time frame. Within our cohort, AVF were successfully created in seven patients after an average duration 407 days on a tunneled catheter. Group A included 16 catheters, which were studied for 4978 days and removed prematurely due to catheter failure. Group B included 29 catheters, studied for 7612 days and were either removed electively or were still functioning at the end of the study. Duration of follow up ranged from 8 to 774 days with a median of 306 ± 28 days. There were no missing data as all the relevant catheter details are routinely entered prospectively into predesigned vascular access forms and stored in the medical records. Three patients were lost to follow up after 172, 456, and 488 days, respectively. Catheters in these three were classified into group A or B according to the catheter status observed until the last follow up.
Patient and catheter characteristics
The age of the children ranged from 5 to 16 years with a mean of 10.16 ± 2.4 years. Congenital anomalies of kidney and urinary tract (CAKUT) was the underlying etiology in 26 (57.8%). The size of the catheter used varied from 8.5 to 14.5 Fr based on the size of the child and catheter availability. The right IJV was the site of catheterization in 80% of cases. Silicone catheters (from Medcomp or Mahurkar, available in sizes 8, 10, 12.5 Fr) were used in all patients in Group A and in 20 (69%) of Group B. Polycarbonate urethane (Bard hemosplit Equistream only in 14.5 Fr) catheters were used in nine (31%) of group B and none in Group A (P = 0.02). The two groups were similar in age, gender, underlying kidney disease, size of catheter used, and site of catheterization [Table 1]
| Group A | Group B | Total | P | |
|---|---|---|---|---|
| Total number of catheters | 16 | 29 | 45 | |
| Age (years) | 9.97±2.3 | 10.28±2.5 | 10.16±2.4 | 0.69 |
| 0-5 yrs, n (%) | 1 (6.2) | 1 (3.4) | 2 (4.4) | 1 |
| 6-10 yrs, n (%) | 9 (56.3) | 13 (44.8) | 22 (48.8) | 0.75 |
| 11-16 yrs, n (%) | 6 (37.5) | 15 (51.7) | 21 (46.6) | 0.76 |
| Gender | ||||
| Boys, n (%) | 13 (81.2) | 25 (82.7) | 38 (84.4) | 0.69 |
| Girls, n (%) | 3 (18.8) | 4 (13.8) | 7 (15.6) | |
| Underlying disease | ||||
| CAKUTa, n (%) | 11 (68.8) | 15 (51.7) | 26 (57.8) | 0.35 |
| Non-CAKUT, n (%) | 5 (31.2) | 14 (48.3) | 16 (42.2) | |
| Type of catheter | ||||
| Silicone, n (%) | 16 (100) | 20 (68.9) | 36 (80) | 0.02 |
| Polyurethane, n (%) | 0 (0) | 9 (31) | 9 (20) | |
| Site of catheter insertion | ||||
| R-.IJVb, n (%) | 13 (81.2) | 23 (79.3) | 36 (80) | 0.99 |
| L-IJVc, n (%) | 3 (18.75) | 6 (20.7) | 9 (20) |
Group A, catheter failure; Group B, elective catheter removal; aCAKUT, Congenital anomalies of kidney and urinary tract; bRIJV, Right internal jugular vein; cLIJV, Left internal jugular vein
Ten (27%) children underwent transplantation by the end of the study with a mean of 293.5 days after initiation of dialysis. Nine (25%) died; three died (8.3%) due to CRBSI associated sepsis and six (16.7%) due to causes unrelated to the catheter.
The median follow up till removal in group A and group B was 338 ± 38 and 593 ± 43 days, respectively. Premature catheter removal in group A was due to CRBSI in 12/16 and mechanical complications in 4/16. The causes of elective catheter removal in group B were kidney transplantation in seven, creation of a AVF in six, change of dialysis modality to peritoneal dialysis in five, and death with a functioning access in four. The remaining seven catheters were functional till the end of the study.
Complications
CRBSI occurred in 15 (33%) catheters with an incidence of 1.19 episodes per 1000 catheter days and led to catheter removal in 12/15 (80%). The incidence of infection in Group A (n = 12) was 2.41 episodes per 1000 catheter days or 0.87 per patient catheter years. In Group B, the incidence of CRBSI was 0.39 episodes per 1000 catheter days (n = 3) [Table 2-I]. Gram-negative bacteria were the causative organisms in 80% of the cases. The organisms causing CRBSI in the two groups were different and are depicted in Table 2-II.
| Group A (16) | Group B (29) | Total (45) | P | |
|---|---|---|---|---|
| (I) Catheter adverse events | ||||
| A) CRBSIa, n (%) | 12 (75) | 3 (10.3) | 15 (33.3) | <0.001 |
| Episodes | 2.41* | 0.39* | 1.19* | |
| B) Thrombosis, n (%) | 8 (50) | 5 (17.2) | 13 (28.8) | 0.04 |
| Episodes | 1.6* | 0.65* | 1.03* | |
| C) Catheter malfunction, n (%) | 4 (25) | 1 (3.4) | 5 (11) | |
| Kink, n (%) | 1 (6.25) | 1 (3.4) | 2 (4.4) | 1.00 |
| Fracture, n (%) | 3 (18.75) | 0 (0) | 3 (6.6) | 0.039** |
| Episodes | 0.8* | 0.13* | 0.39* | |
| (II) Organisms isolated in catheter-related infections (15) | ||||
| NFGNBb | 0 | 2 (66.6) | 2 (13.3) | N/A |
| Klebsiella | 0 | 1 (33.3) | 1 (6.6) | N/A |
| Pseudomonas | 6 (50) | 0 | 6 (40) | N/A |
| MRSAc | 2 (16.6) | 0 | 2 (13.3) | N/A |
| Citrobacter | 2 (16.6) | 0 | 2 (13.3) | N/A |
| Acinetobacter | 1 (8.3) | 0 | 1 (6.6) | N/A |
| Candida glabrata | 1 (8.3) | 0 | 1 (6.6) | N/A |
Group A, catheter failure; Group B, elective catheter removal; aCRBSI, catheter-related blood stream infections; bNFGNB; Non fermenter gram negative bacilli, cMRSA; Methicillin-resistant staphylococcus aureus.*Expressed as episodes per 1000 catheter days,**Fisher exact statistic value
Of the 12 infected catheters in Group A, 6 were changed to a HD catheter at a different site and 3 were shifted to peritoneal dialysis. Three patients died due to sepsis-related complications. All infected catheters in group B were salvaged.
Thrombus was seen in 13 catheters, in 8/16 (50%) of the catheters in group A, with an incidence of 1.6 episodes/1000 catheter days as opposed to 5/29 (17.2%) of the catheters in group B, with an incidence of 0.52 episodes/1000 catheter days. Of 13 episodes of thrombus, 9 were associated with CRBSI [Table 2-I]. In group A, five catheters responded to thrombolytic treatment, while three required removal due to non-resolution. Group B showed resolution with treatment in all four catheters. Mechanical complications occurred in group A in four with kink in one and fracture in three. There was one kink in group B. [Table 2-I]
Risk factors for CRBSI
No specific patient characteristic could be identified as a risk factor for CRBSI. Among the catheter characteristics, the insertion site, catheter size, and type had no significant association with CRBSI. Binary logistic regression analysis showed that the presence of thrombus and duration of catheterization were significant predictors of infection. The presence of a thrombus had an OR of 29.51 (95% CI 3.5, 248), P = 0.009 and longer duration of catheterization had an OR of 1.009 per day (95% CI 1.002, 1.016), P = 0.002. [Table 3].
| Catheters with infection | Catheters without infection | Relative risk (95% C.I.) | P | |
|---|---|---|---|---|
| Number of catheters | 15 | 30 | ||
| Age-Mean±SD (yrs) | 10.46±2.69 | 10.01±2.33 | 0.35 | |
| Distribution | ||||
| <5 yrs, n (%) | 1 (6.6) | 1 (3.3) | 1.5 (0.3,3.6) | 0.76 |
| 6-10 yrs, n (%) | 8 (53.3) | 14 (46.6) | 1.1 (0.5,2.7) | |
| >11 yrs, n (%) | 6 (40) | 15 (50) | 0.86 (0.35,2) | |
| Gender | ||||
| Boys, n (%) | 11 (73.3) | 27 (90) | 0.5 (0.24,1.3) | 0.15 |
| Girls, n (%) | 4 (26.6) | 3 (10) | ||
| Underlying disease | ||||
| CAKUTa | 10 (66.6) | 21 (70) | 0.9 (0.4,2.2) | 0.82 |
| Non-CAKUT | 5 (33.3) | 9 (30) | ||
| Size of catheter | ||||
| <12.5 Fr | 5 (33.3) | 16 (53.3) | 0.57 (0.22,1.34) | 0.2 |
| ≥12.5 Fr | 10 (66.6) | 14 (46.6) | ||
| Site of catheter | ||||
| R-IJVb, n (%) | 12 (80) | 24 (80) | 1 (0.42,3.02) | 1 |
| L-IJVc, n (%) | 3 (20) | 6 (20) | ||
| Type of catheter | ||||
| Silicone, n (%) | 14 (93.3) | 22 (73.3) | 3.5 (0.8,20.47) | 0.23 |
| Polyurethane, n (%) | 1 (6.6) | 8 (26.7) | ||
| Prior hemodialysis catheter inserted at same site, n (%) | 4 (26.6) | 10 (33.3) | 0.81 (0.29,1.9) | 0.19 |
| Presence of thrombosis, n (%) | 9 (69.2) | 4 (13.3) | 29.5 (3.5,248.5)** | 0.002 |
| Duration of catheterization (median days, IQR) | 362 (316,450.5) | 187.5 (81,339.3) | 1.009 (1.002,1.009)** | 0.009 |
| Catheter failure, n (%) | 12 (80) | 4 (10) | 8 (2.9,23.9) | <0.001 |
aCAKUT, Congenital Anomalies of Kidney and Urinary tract; bR-IJV, Right Internal jugular vein; cL-IJV, Left internal jugular vein, **Odds ratio (95% C.I.) derived from the binary regression
The construction of the ROC curve showed an area under the curve of 0.808 for duration of catheterization as a predictor for risk of CRBSI and identified 267 days as the cut-off point with a sensitivity of 93% and a specificity of 66.7% [Figure 2]. Infection free catheter survival for the group was a mean of 449 ± 42 days with 65% infection free survival at 360 days. The infection free survival at 360 days was 50% for group A vs 84.2% for group B (P = 0.03) [Figure 3].
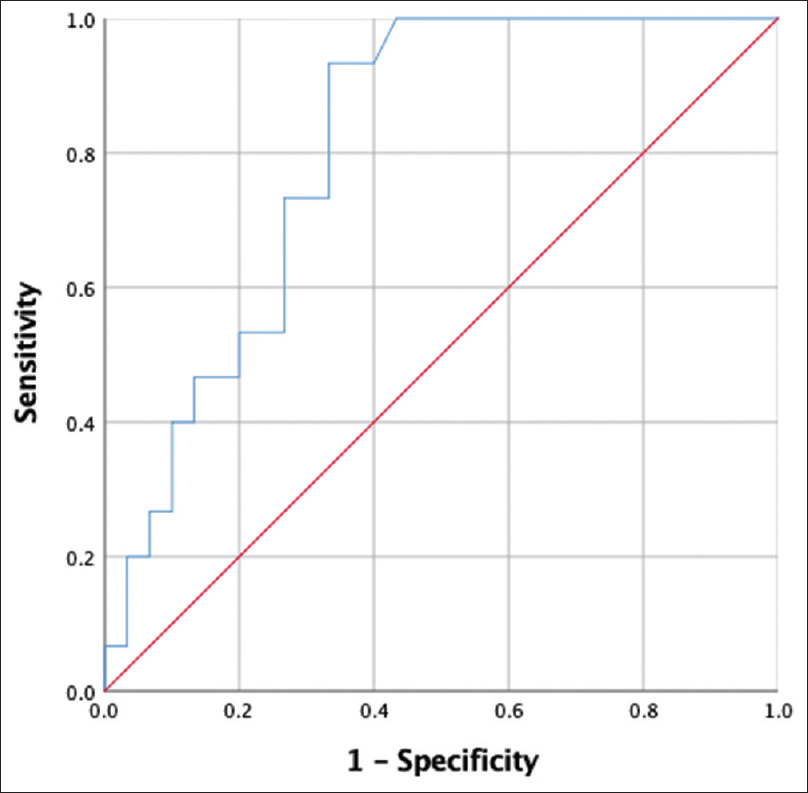
- ROC curve predicting a cut-off value of catheter duration for infection. area under the curve = 0.808 (P = 0.001), determined a cutoff for the duration of catheterization to predict infection to be 267 days. (Sensitivity = 93%, Specificity = 66.7%)
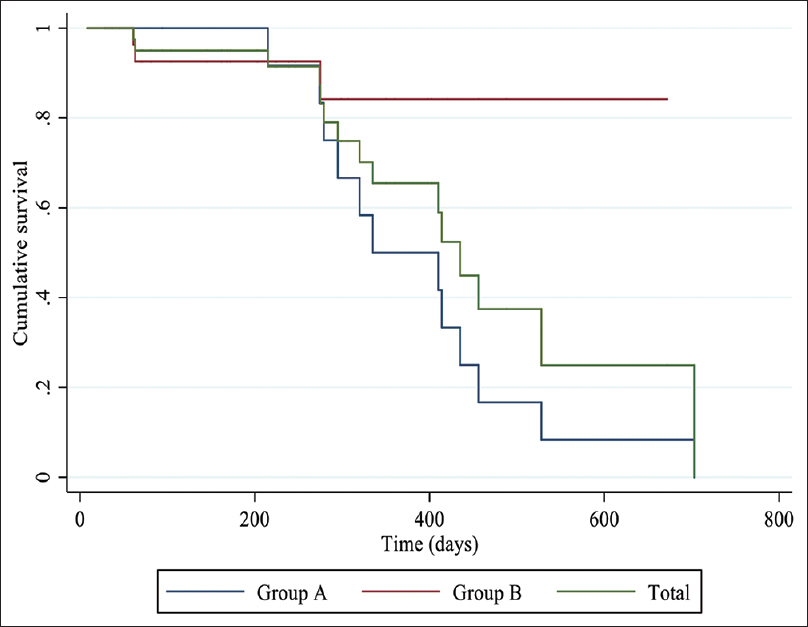
- Kaplan– Meir charts comparing infection-free survival in catheter failure (group A), elective removal (group B), and cumulative group. KM curves showing cumulative survival of the total group (green) showing mean survival of 449 ± 42 days with survival 95%@90 and 180 days, 91.5%@ 270 days, and 65.5% @ 360 days; KM curves showing infection free survival of group A (blue) with mean survival of 388 ± 38 days with 100% survival at 90 and 180 days, 91.7% @270 days, and 50% @360 days and group B (red) with mean survival of 593 ± 43 days with 92.6% @ 90,180, and 270 days, 84.2% @360 days. (P = 0.03)
Discussion
Median catheter survival in our cohort was 306 ± 28 days. Catheter failure, defined as unplanned premature removal due to catheter-associated complications, occurred in one-third of all catheters and CRBSI being responsible for three-fourths of catheter failures. The predominant organisms isolated in our patients were gram-negative bacteria accounting for 80% of all organisms, half of which were due to pseudomonas. Staphylococci accounted for only two cases in our study. Thrombus within the catheter was seen in more than two-thirds of the catheters with CRBSI and had a high predictive value for the presence of CRBSI. The overall mortality in the cohort was 25% with 19% attributed to sepsis related to CRBSI. Infection free catheter survival was greater than 90% during the first 270 days but dropped to 65% by 360 days. The duration of catheterization of 267 days was identified as a significant cut-off for the risk of CRBSI.
The CRBSI rate in our study was 1.19 per 1000 catheter days, which was comparable to those reported in many other studies [Table 4].[21314] In the study by Goldstein et al.,[24] the CRBSI rate was 0.71 per patient year and was the cause for catheter failure in 36% of cases with no related mortality. The low prevalence had been attributed to strict adherence to aseptic protocols. Mortality secondary to CRBSI was higher (1.6%) than reported in other studies,[25] probably due to nature of causative organism. Similar gram-negative predominance has been shown only in one of the adult studies from South Asia with 40% resistant strains.[26] On the contrary, most pediatric studies have reported staphylococci to be the most frequent bacterial isolate.[112728]
| Author (published) | Site | Median age | Catheters (N)/patients | CRBSIb | Thrombusb | Mechanicalb | Median survival (range) |
|---|---|---|---|---|---|---|---|
| Borzych-Duzalka et al. (2019)[2] | Europe | 11.1 yrs (6.5,14.7) | 628 TCCs/404 | 1.3 | 2.5 (combined) | 420 days | |
| Keenswijk et al.(2018)[14] | Africa | 52±4.2 monthsa | 21 TCCs/15 | 1.1 | 3.7 | NR | 132 days (3,1470) |
| Paglialonga et al.d (2016)[12] | Italy | 11.4 months | 18 TCCs/16 | 0.6 | 0.6 | NR | 349 days |
| Wang et al. (2015)[9] | China | 10±4.2 yrs | 21 TCCs/16 | 6.9 (3.7) | 4.6 | 1.6 | 420±66 days (9.6,18.3) |
| Lopez et al. (2014)[13] | Chile | 27.5 monthsa | 31 TCCs/11 | 1.37c | 1.7c | 2.4c | 312 daysa (26,840) |
| Eisentein et al. (2011)[31] | Israel | 10 yrs (0.25,17) | 59 TCCs/29 | 0.52 | NR | NR | 310 days (27,1578) |
| Current study | India | 10.16±2.4 yrs | 45 TCCs/36 | 1.19 | 1.03 | 0.39 | 306±28 days (250,362) |
aUsing mean values, bValues determined as incidence per 1000 catheter days, cIncidence leading to catheter failure; dmulticentric studies; TCC, tunelled cuffed catheter; CRBSI, catheter-related blood stream infections
No patient characteristic or catheter characteristic could be identified as a risk factor for CRBSI. Though young age has been reported as risk factor in most studies, the Italian registry along with few others showed a lower prevalence of CRBSI in children less than 2 years of age.[12293031] Our study had only two children aged less than 5 years, making this finding difficult to assess. Hypoalbuminemia and immunosuppression have been identified as risk factors for CRBSI in some studies.[303233] Although we did not specifically address these issues, the incidence of CRBSI was similar in those with CAKUT and non-CAKUT as the underlying primary renal disease. Catheter-related risk factors included the site of catheter insertion, with femoral vein and left IJV being more susceptible.[34] Other factors incriminated were duration of catheterization and past catheter-related bacteraemia.[323536] Eighty percent of our patients had a right IJV insertion, and none with femoral access; hence, we could not assess relevance of site of insertion. No significant difference was found in incidence of CRBSI based on the type of catheter used, but only one-ninth Bard catheters developed CRBSI and none of them failed. This catheter was available only in larger size (14.5 Fr) and hence used only in older children. However, the numbers are too few to assess its significance.
A positive correlation between mural thrombi and infection had also been noted by Raad et al.[37] However, the temporal relationship between the thrombus and CRBSI could not be ascertained due to the retrospective nature of the study. We did not come across a fibrin sheath leading to obstruction.
The safe duration of catheterization has not been addressed in many studies, but similar critical cut off of 9 months was shown by Goldstein et al.[24] Hence, in lieu to the cut off in our study of 267 days, early AVF formation can prevent the infectious complications. Mechanical complications accounted for one-fourth of all catheter failure in our patients, though have been significantly higher accounting for 23–81% of the catheter failures.[10121331]
Indian children with ESRD who do not have an eligible kidney donor in the family have a long dialysis vintage, as the waiting period for cadaveric donation can be longer than 5 years. The survival to transplantation is dependent not only on patient characteristics and access to good nephrology care but also on the survival of the tunneled catheter. Mortality is known to be significantly higher in children on dialysis than in those with transplantation.[438] In developed countries that have had maintenance HD for several decades, a reduction in mortality has been seen after each successive 5-year period with many countries having a mortality rate less than 5% for children on maintenance dialysis.[39404142] In contrast, high mortality rates greater than 20% has been reported from many resource limited nations including India.[1543] Constraints in India include the lack of trained nurses and technicians for pediatric dialysis, late diagnosis, poor nutritional state of the patients, and the need to self-pay for dialysis care at many centers.
There are several limitations of our study. It is a single center study with a small sample size, which limits the generalizability of results. The study is retrospective in nature with its inherent bias and inability to establish a temporal relationship of risk factors. Other long-term complications of tunneled catheters such as fibrin sheath formation, central vein stenosis, access recirculation, and flow limitations that may adversely impact the quality of dialysis were not addressed in our study. Our study could not identify significant patient or catheter-related risk factors for catheter failure, probably because of the small sample size and retrospective nature of the study. Prospective large studies may help to identify other risk factors for CRBSI (like age, thrombosis, site) and define the role of different catheter types in the prevention of infection.
In conclusion, despite the significant risk of adverse events, tunneled catheters remain a viable option for maintenance HD in children, even in resource limited settings, as 75% of children survived at the end of 6 years with successful transplantation in one-fourth of the children. CRBSI is the main reason of catheter failure. Early creation of A-V fistula must be a reasonable necessity aiming at preventing infectious complications.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Bibliography
- Challenges of long-term vascular access in pediatric hemodialysis: Recommendations for practitioners. Hemodial Int. 2021;25:3-11.
- [Google Scholar]
- Vascular access choice, complications, and outcomes in children on maintenance hemodialysis: Findings from the international pediatric hemodialysis network (IPHN) registry. Am J Kidney Dis. 2019;74:193-202.
- [Google Scholar]
- US renal data system 2019 annual data report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2020;75((Suppl 1)):A6-7.
- [Google Scholar]
- US Renal Data System 2018 Annual Data Report: Epidemiology of kidney disease in the United States. Am J Kidney Dis. 2019;73((3S1)):A7-8.
- [Google Scholar]
- Tunneled catheters in hemodialysis patients: Reasons and subsequent outcomes. Am J Kidney Dis. 2005;46:501-8.
- [Google Scholar]
- Management of hemodialysis catheter-related bacteremia with an adjunctive antibiotic lock solution. Kidney Int. 2002;61:1136-42.
- [Google Scholar]
- Predictors and outcome of catheter-related bacteremia in children on chronic hemodialysis. Pediatr Nephrol. 2006;21:1452-8.
- [Google Scholar]
- Prevention and management of catheter-related infection in hemodialysis patients. Kidney Int. 2011;79:587-98.
- [Google Scholar]
- Cuffed-tunneled hemodialysis catheter survival and complications in pediatric patients: A single-center data analysis in China. Int J Clin Exp Med. 2015;8:9765-71.
- [Google Scholar]
- Pediatric hemodialysis: Lessons from the past, ideas for the future. Kidney Int Suppl. 1996;53:S64-7.
- [Google Scholar]
- Management of hemodialysis catheter-related bacteremia –A 10-year experience. Pediatr Nephrol. 2000;14:198-202.
- [Google Scholar]
- Chronic haemodialysis in small children: A retrospective study of the Italian Pediatric Dialysis Registry. Pediatr Nephrol. 2016;31:833-41.
- [Google Scholar]
- Outcome of tunnelled central venous catheters used for haemodialysis in children weighing less than 15 kg. J Pediatr Surg. 2014;49:1300-3.
- [Google Scholar]
- Audit of hemodialysis in children weighing less than 20 kg in an African pediatric nephrology unit. Ther Apher Dial. 2018;22:617-23.
- [Google Scholar]
- Management of pediatric end stage kidney disease in a developing country-change in the trend over 12 years. Kidney Int Rep. 2020;5:S281.
- [Google Scholar]
- Etiology and outcome of chronic renal failure in Indian children. Pediatr Nephrol. 1999;13:594-6.
- [Google Scholar]
- Differential time to positivity: A useful method for diagnosing catheter-related bloodstream infections. Ann Intern Med. 2004;140 doi:10.7326/0003-4819-140-1-200401060-00007
- [Google Scholar]
- Differential time to positivity of blood cultures: A valid method for diagnosing catheter-related bloodstream infections in the intensive care unit. Med Intensiva. 2012;36:169-76.
- [Google Scholar]
- Interventional nephrology: Catheter dysfunction- prevention and troubleshooting. Clin J Am Soc Nephrol. 2013;8:1234-43.
- [Google Scholar]
- KDOQI clinical practice guideline for vascular access:2019 Update. Am J Kidney Dis. 2020;75((4 Suppl 2)):S1-164.
- [Google Scholar]
- Treatment of long-term intravascular catheter-related bacteraemia with antibiotic-lock therapy. J Antimicrob Chemother. 2006;58:816-21.
- [Google Scholar]
- Hemodialysis tunneled catheter noninfectious complications. Can J Kidney Heal Dis. 2016;27:2054358116669130.
- [Google Scholar]
- Recombinant urokinase for restoration of patency in occluded central venous access devices. A double-blind, placebo-controlled trial. Thromb Haemost. 2004;92:575-82.
- [Google Scholar]
- Hemodialysis catheter survival and complications in children and adolescents. Pediatr Nephrol. 1997;11:74-7.
- [Google Scholar]
- National healthcare safety network (NHSN) dialysis event surveillance report for 2014. Clin J Am Soc Nephrol. 2017;12:1139-46.
- [Google Scholar]
- Fast and furious: A retrospective study of catheter-associated bloodstream infections with internal jugular nontunneled hemodialysis catheters at a tropical center. Clin Kidney J. 2019;12:737-44.
- [Google Scholar]
- Survival and complications of cuffed catheters in children on chronic hemodialysis. Pediatr Nephrol. 1999;13:245-8.
- [Google Scholar]
- Bacteremia in a pediatric hemodialysis unit secondary to Enterococcus fecalis. Pediatr Nephrol. 1996;10:55-7.
- [Google Scholar]
- Catheter survival and comparison of catheter exchange methods in children on hemodialysis. Pediatr Nephrol. 2007;22:1355-61.
- [Google Scholar]
- Vascular access survival in children and young adults receiving long-term hemodialysis. Am J Kidney Dis. 2005;45:708-14.
- [Google Scholar]
- Low infection rates and prolonged survival times of hemodialysis catheters in infants and children. Clin J Am Soc Nephrol. 2011;6:793-8.
- [Google Scholar]
- Current concepts in hemodialysis vascular access infections. Adv Chronic Kidney Dis. 2019;26:16-22.
- [Google Scholar]
- Vascular access: Choice and complications in European paediatric haemodialysis units. Pediatr Nephrol. 2012;27:999-1004.
- [Google Scholar]
- Tunneled internal jugular hemodialysis catheters: Impact of laterality and tip position on catheter dysfunction and infection rates. J Vasc Interv Radiol. 2013;24:1295-302.
- [Google Scholar]
- Dialysis catheter-related bacteremia:treatment and prophylaxis. Am J Kidney Dis. 2004;44:779-91.
- [Google Scholar]
- Natural history of tunneled dialysis catheters placed for hemodialysis initiation. J Vasc Interv Radiol. 2013;24:1289-94.
- [Google Scholar]
- The relationship between the thrombotic and infectious complications of central venous catheters. JAMA. 1994;271:1014-6.
- [Google Scholar]
- Survival advantage of pediatric recipients of a first kidney transplant among children awaiting kidney transplantation. Am J Transplant. 2008;8:2600-06.
- [Google Scholar]
- Mortality risk among children initially treated with dialysis for end-stage kidney disease, 1990-2010. JAMA. 2013;309:1921-9.
- [Google Scholar]
- Available from: https://www.sciencedaily.com/releases/2013/05/130508131823.htm
- Mortality risk in European children with end-stage renal disease on dialysis. Kidney Int. 2016;89:1355-62.
- [Google Scholar]
- Preemptive kidney transplantation is associated with survival benefits among pediatric patients with end-stage renal disease. Kidney Int. 2016;90:1100-8.
- [Google Scholar]
- Management and outcome of children with end-stage renal disease in northwest Iran. Indian J Nephrol. 2012;22:94-7.
- [Google Scholar]







