Translate this page into:
Clinicopathological Spectrum of Biopsy Proven Kidney Disease in Diabetics
Corresponding author: Shyam Bihari Bansal, Department of Nephrology, Medanta Institute of Kidney and Urology, Medanta-The Medicity, Gurugram, Haryana, India. E-mail: drshyambansal@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mittal A, Rana AS, Lokkur PP, Manhas N, Sethi SK, Singh V, et al. Clinicopathological Spectrum of Biopsy Proven Kidney Disease in Diabetics. Indian J Nephrol. doi: 10.25259/IJN_699_2024
Abstract
Background
Diabetes mellitus is the leading cause of chronic kidney disease worldwide. Nephrologists might miss nondiabetic kidney diseases (NDKD) because they might avoid biopsies due to the presumption of diabetic kidney disease (DKD). This study retrospectively analyzed kidney biopsies of patients with diabetes in a large Indian multi-super specialty tertiary care private hospital.
Materials and Methods
Case records of 130 diabetic patients who underwent kidney biopsies at the institute between January 2013 and December 2022 were included.
Results
The mean age was 56 ± 9.7 years. There were 102 (78.4%) males; 79 (60.79%) had NDKD, 34 (26.15%) had DKD, and 17 (13.07%) had combined disease. Diabetic retinopathy was more common in those with DKD (58.5%) than NDKD (8.8%) (P=<0.001). The mean duration of diabetes in NDKD was 7.92 ± 4.14 years, compared to 9.44 ± 5.51 and 9.77 ± 5.54 years in those with DKD and combined disease, respectively (p=0.27). Nephrotic syndrome was most common, followed by rapidly progressive renal failure and acute kidney injury. The most common glomerular disease with NDKD, diagnosed by kidney biopsy, was membranous nephropathy (13.92%), followed by IgA Nephropathy (12.66%), and focal segmental glomerulosclerosis (8.86%).
Conclusion
NDKD was seen in most patients with diabetes who underwent a kidney biopsy. For patients with diabetes presenting atypical features, a low threshold for kidney biopsy might change the management and outcomes.
Keywords
Chronic kidney disease
Diabetic kidney disease
Diabetes mellitus
Kidney biopsy
Nondiabetic kidney disease
Introduction
Diabetes mellitus (DM) is the leading cause of chronic kidney disease (CKD) worldwide.1 The number of diabetes cases is expected to increase from ∼537 million in 2021 to 643 million by 2030.2 With 30-40% of patients with DM developing CKD, it is one of the most devastating complications. Diabetic kidney disease (DKD) accounts for ∼40% of all patients with CKD.3 It usually occurs after long-term diabetes and is diagnosed on clinical ground with gradually increasing proteinuria, edema, worsening hypertension (HTN), and renal dysfunction. Most patients with DKD have associated diabetic retinopathy (DR) due to long-standing history and often poor diabetes control. Sometimes, patients with DM may also have nondiabetic kidney diseases (NDKD), which could be missed if not adequately evaluated. Studies have reported variable NDKD incidence. Prior cohorts of native kidney biopsy in patients with diabetes showed ∼65-70% of patients with DM with NDKD.4-6 A study also showed that biopsies of 25-50% of patients with type 2 diabetes showed glomerular lesions unrelated to or in addition to DKD after biopsy.7 Variations in selection criteria, study design, reporting bias, and geographic and ethnic differences may have caused the wide range and discrepancies in these studies. NDKD can occur in isolation or coexist with diabetic nephropathy (DN). Timely NDKD diagnosis in patients with DM becomes essential as it could be reversible and have a good prognosis. Hence, in patients with DM showing atypical presentation having a high index of suspicion of NDKD, performing a kidney biopsy is essential. In this study, we reviewed the biopsies of patients with DM who presented with atypical features like sudden nephrotic syndrome (NS) onset, active urinary sediments, acute nephritic syndrome, acute kidney injury (AKI), and rapidly progressive renal failure (RPRF).
Materials and Methods
This retrospective study was conducted at Medanta - The Medicity, Gurugram in India. The case records of patients >18 years with diabetes who underwent kidney biopsy at our center between January 2013 and December 2022 were taken from the hospital information system. The demographic findings, biopsy indication, and clinical and laboratory data were reviewed. Since this was a retrospective observational analysis of the compiled data, it was exempted from ethics committee clearance of our institute and taking consent was thus skipped. During the whole process of research, confidentiality and privacy of the patients were assured.
The analyzed clinical variables estimated by CKD epidemiology and kidney biopsy included duration of diabetes, age, gender, presence of systemic HTN, hemogram, serum albumin, serum creatinine, and estimated glomerular filtration rate (eGFR). Proteinuria was quantified using the urine albumin creatinine ratio (UACR) in a spot sample or 24-hour urine protein estimation. Microscopic hematuria was defined as >3 RBCs per high power field on the most recent urine analysis before kidney biopsy. All patients underwent fundus examination for DR. Patients were excluded if pathology reports indicated malignancy or if there was insufficient tissue (i.e., lack of sufficient tissue to make a concrete diagnosis).
Based on clinical and laboratory data, clinical presentations were defined as NS, nephritic syndrome, AKI, RPRF, or acute or CKD. Kidney diseases were categorized as isolated DN, isolated NDKD, and NDKD with DN (combined disease). NDKD patients were further divided as having primary, secondary, and tubulointerstitial glomerulopathies.
Kidney biopsies were performed with real-time ultrasound guidance by a nephrologist with a 16-gauge needle. Sections for light microscopy were stained with hematoxylin and eosin, Schiff’s periodic acid, Methenamine silver, and Masson’s trichrome. Immunofluorescence staining was done with antibodies against IgG, IgA, IgM, C3 and C1q, ĸ, and λ light chains. Electron microscopy was performed in selected cases.
Continuous data were presented as the mean and standard deviation, and categorical variables as number and percentage. Data were analyzed with one-way ANOVA (Welch’s test) and Kruskal-Wallis test. Multinominal logistic regression was used to estimate the odds ratio, associated 95% confidence interval.
Results
During the study, 1460 patients underwent kidney biopsy at our center, of which 132 had DM. Two were excluded due to insufficient kidney tissue samples. The mean age was 56 ± 9.7 years with a range of 29 - 81 years; there were 102 males and 28 females. At presentation, there was no difference in most baseline characteristics among groups, including age, duration of diabetes, proteinuria, hemoglobin level, serum albumin, and serum creatinine. Of 130 patients, 98 (75.38%) had HTN and 35 (26.92%) had DR. The median spot UACR was 3.61 g/day, with a range of 0.06 g/day to 25.13 g/day; the groups had no difference in UACR values. (p = 0.501) [Table 1].
| Characteristics | NDKD (class 1) | DN (class 2) | NDKD + DN (class 3) | P value |
|---|---|---|---|---|
| Patient (n) | 79 | 34 | 17 | |
| Age (years) | 54.6 ± 9.41 | 55.5 ± 10.7 | 60 ± 9.1 | 0.104 |
| Female | 23 | 5 | 0 | <0.001 |
| Duration of diabetes | ||||
| <8 Years | 52 (65.8%) | 15 (44.1%) | 6 (35.3%) | 0.09 |
| >8 Years | 27 | 19 | 11 | |
| Mean duration of diabetes (years) | 7.93 ± 4.14 | 9.44 ± 5.51 | 9.77 ± 5.54 | 0.27 |
| Patients on antidiabetic drugs | 75 | 33 | 17 | |
| Patients on RAASi (n=81)* | 26 (n=49) | 12 (n=20) | 5 (n=12) | |
| Renal dysfunction | 64 | 31 | 14 | 0.40 |
| Serum creatinine (mg/dL) | 3.93 ± 2.95 | 3.61 ± 2.35 | 3.43 ± 1.78 | 0.996 |
| Serum albumin (mg/dL) | 2.91 ± 0.63 | 3.08 ± 0.68 | 3.23 ± 0.81 | 0.248 |
| Hemoglobin (g/dL) | 10.7 ± 2.11 | 10.3 ± 1.76 | 10.3 ± 1.76 | 0.526 |
| Median UACR (range) | 3.5 (0.06-25) | 4.2 (0.17 -20) | 3.48 (0.23 -25.13) | 0.501 |
| Microscopic hematuria (>3rbc/hpf) | 30 | 5 | 3 | <0.001 |
| Proteinuria | 67 | 30 | 13 | 0.548 |
| Number of glomeruli on light microscopy | 14 ± 7.57 | 16 ± 9.3 | 17 ± 5.6 | 0.266 |
| Number of glomeruli on immunofluorescence microscopy | 7 ± 4.33 | 6 ± 1.55 | 8 ± 4.8 | 0.047 |
| Interstitial fibrosis and tubular atrophy (%) | 15.8 ± 15.9 | 34.3 ± 17.7 | 21.8 ± 16.8 | <0.001 |
| Diabetic retinopathy | 7 | 20 | 8 | <0.001 |
DN: Diabetic nephropathy, NDKD: Nondiabetic kidney disease, UACR: Urine albumin/creatinine ratio, RBC: Red blood cells, hpf: High power field, RAAS: Renin angiotensin aldosterone system inhibitor, *as study was retrospective, data for patients on RAASi was available only for 81 patients. Bold value indicates statistical significance.
Amongst those with diabetes for <8 years, a statistically significant difference was not seen between patients NDKD (isolated or combined) and DN (65.8% vs 35.3% vs 45.1%; p=0.09). NDKD had a greater predilection for females than DKD (29.11% vs 14.7%; p-value = <0.001; odds ratio = 1.789; 95% CI = 1.785- 1.792). At presentation, there was no difference in the mean eGFR values of patients with NDKD and DN (26 ± 23.64 vs 29 ± 20.86 mL/min/1.73m2; p-value = 0.944). Retinopathy was less common in patients with NDKD than those with DKD (8.8% vs 58.8%; p = <0.001; odds ratio = 0.047; 95% CI = 0.0469 - 0.0471).
Patients with DN and DR had an association with sensitivity and specificity of 54.9% (95%CI = 40.34% to 68.87%) and 91.14% (95%CI = 82.59% to 96.36%), respectively. The positive (PPV) and negative predictive values (NPV) were 80% (95%CI = 65.4% to 89.44%) and 75.79% (95%CI = 69.65% to 81.03%), respectively.
Microscopic hematuria was more common in patients with NDKD than those with DN or combined disease (37.97% vs 14.7% vs 17.64%; p = <0.001). Kidney biopsy showed 79 (60.79%) patients with isolated NDKD, 34 (26.15%) with isolated DN, and 17 (13.07%) patients with NDKD and DN features together. Other baseline characteristics have been shown in Table 1.
The most common clinical presentation was NS (36.92%), followed by AKI (19.23%) and RPRF (18.46%) [Figure 1 and Table 2]. Renal dysfunction without proteinuria and acute nephritic syndrome were other presentations.
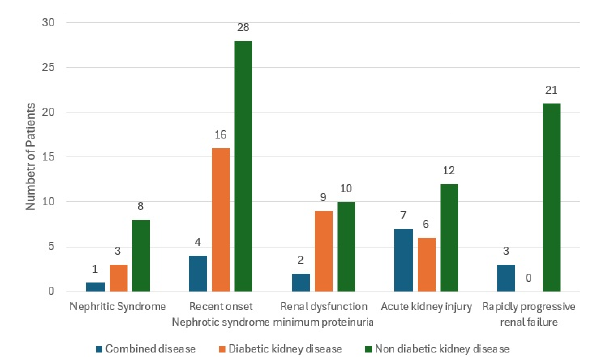
- Indication for kidney biopsy in patients with type 2 diabetes mellitus.
| Indications of kidney biopsy | Diabetic kidney disease | Non diabetic kidney disease | Combined disease |
|---|---|---|---|
| Nephritic syndrome | 3 | 8 | 1 |
| Recent onset nephrotic syndrome | 16 | 28 | 4 |
| Renal dysfunction minimum proteinuria | 9 | 10 | 2 |
| Acute kidney injury | 6 | 12 | 7 |
| Rapidly progressive renal failure | 0 | 21 | 3 |
Class III and class IV of renal pathology society diabetic Nephropathy were seen in patients with isolated diabetic nephropathy as compared to combined disease (DKD with NDKD). Similarly, hypertensive nephropathy and interstitial changes were seen more in patients with isolated DN than combined disease [Table 3].
| Kidney biopsy finding | DN (34) | DN + NDKD (17) |
|---|---|---|
| Class I | 0 | 2 |
| Class IIa | 3 | 10 |
| Class IIb | 10 | 5 |
| Class III | 13 | 0 |
| Class IV | 8 | 0 |
| Number of patients with changes in hypertensive nephropathy | 34 | 11 |
| Number of patients with interstitial changes | 32 | 3 |
DN: Diabetic nephropathy, DN + NDKD: Diabetic nephropathy with Non diabetic kidney disease
Membranous nephropathy (MN) (13.92%), IgA nephropathy (12.66%), and focal segmental glomerulosclerosis (8.86%) were the most common glomerular diseases in patients with NDKD. There were four patients with lupus nephritis, amyloidosis, and minimal change disease. Acute tubulointerstitial nephritis was seen in 24.05% of the cases [Table 4]. It was the most common finding in patients of DN with NDKD (41.18%), followed by MN (17.65%) and chronic tubulointerstitial nephritis (11.76%) [Table 4].
| Renal biopsy finding | Total (N = 96) | NDKD (N = 79) | NDKD and DN (N = 17) |
|---|---|---|---|
| ATIN | 26 (27.08%) | 19 (24.05%) | 7 (41.17%) |
| Membranous nephropathy | 14 (14.58%) | 11 (13.9%) | 3 (17.6%) |
| IgA nephropathy | 10 (10.41%) | 10 (12.6%) | |
| FSGS | 8 (8.33%) | 7 (8.8%) | 1 (5.88%) |
| AAV/Pauciimmune | 7 (7.29%) | 6 (7.5%) | 1 (5.88%) |
| Minimal change disease | 5 (5.20%) | 4 (5.06%) | 1 (5.88%) |
| Amyloidosis | 4 (4.16%) | 4 (5.06%) | |
| Lupus nephritis | 4 (4.16%) | 4 (5.06%) | |
| IRGN | 3 (3.12%) | 2 (2.53%) | 1 (5.88%) |
| CTIN | 3 (3.12%) | 1 (1.27%) | 2 (11.76%) |
| Anti-GBM disease | 2 (2.08%) | 2 (2.53%) | |
| Others | 9 (9.37%) | 8 (10.12%) | 1 (5.88%) |
ATIN: Acute tubulointerstitial nephritis, FSGS: Focal segmental glomerulosclerosis, AAV: ANCA associated vasculitis, IRGN: Infection related glomerulonephritis, CTIN: Chronic tubulointerstitial nephritis, GBM: Glomerular basement membrane, NDKD: Non diabetic kidney disease, DKD: Diabetic kidney disease.
The percentage of interstitial fibrosis and tubular atrophy was significantly higher in biopsies of patients with DN and combined disease than those with NDKD (15.8% ± 15.9% vs 34.3% ± 17.7%; p = <0.001).
Discussion
This study demonstrates NDKD’s prevalence and clinicopathological profile in patients with type 2 DM and indications for performing a kidney biopsy in diabetic patients. The reported NDKD prevalence in patients’ biopsies ranges from 3% to 82%.5,8-11 This wide variation is due to different criteria and protocols used at various centers. This study included kidney biopsies of patients presenting atypical features like sudden NS onset, presence of microscopic hematuria, rapid deterioration of kidney functions, absence of retinopathy, and clinical features suggestive of systemic disease. Most patients who underwent kidney biopsies had NDKD, highlighting that in patients with DM, clinical suspicion is able to predict cases of NDKD.
The mean age of the patients with NDKD was 55 ± 9.3 years, which was like other studies.9,12 There was no significant difference in the age and mean diabetes duration between DN and NDKD patients. The mean duration of diabetes was shorter in patients with NDKD compared to those with DN and combined disease, but this difference was not statistically significant. Another study reported similar results where the mean age and duration of diabetes in NDKD were lower, but not statistically significant, than in patients with DKD or combined disease.13 Pham et al. did not show a statistically significant difference in age at the time of biopsy in patients with DKD, NDKD, or combined disease.5 Patients were also stratified as per the median 8 year duration. Patients with duration <8 years did not show a statistically significant difference between isolated NDKD and combined disease with DKD. Hence, our observations suggest that the age of the patients and the duration of diabetes are not sensitive indicators differentiating between DN and NDKD. NDKD was significantly more prevalent in females than isolated DN. In contrast, Kritmetapak et. al., showed a higher tendency of NDKD in males.9 However, the number of women in our cohort was limited, so it is difficult to make any conclusion on the effect of gender. The median UACR between DKD and NDKD groups was not significantly different. This finding is supported by most previous studies. Patients with DN had statistically significant proteinuria.14-16 Hematuria was significantly more common in patients with NDKD than those with DN or combined disease. This presence or absence is not a sensitive DN indicator, and kidney biopsy should be done irrespectively for patients with high clinical suspicion of NDKD.
Isolated DN and NDKD were more present in the presence and absence of retinopathy, respectively. Only 60% of patients with biopsy-proven DN had retinopathy. There is a higher NPV for DR as most patients with NDKD did not have it. The sensitivity and specificity of DR for isolated DN were 54.9% and 91.14%, respectively. The PPV and NPV were 80% and 75.79%, respectively. He et al. calculated the sensitivity and specificity of DR for isolated DN as 65% and 75%, respectively.17 This also indicates that the absence of retinopathy does not exclude DN.
NS was the most common presentation, followed by AKI and RPRF. Different studies observed and reported a similar heterogeneous kidney diseases presentation in diabetics.13,17-19
In this study, MN and IgA nephropathy were the most common nondiabetic glomerular lesions seen on kidney biopsy. These lesions are potentially treatable with medications other than ACE inhibitors, angiotensin receptor blockers, or SGLT2 inhibitors.20,21 Studies from different centers reported similar prevalences of different glomerular diseases in diabetic patients.10,22-24 Few studies have demonstrated IgA nephropathy as the most common finding in kidney biopsy.9,14,16,25
Interstitial and tubular involvement in the form of IFTA was significantly higher among patients with isolated DN or combined disease as compared to those with NDKD. This suggests a higher degree of chronicity among patients with DN or combined disease. Other studies also reported a higher degree of chronicity in the form of IFTA among patients with DN than NDKD.24
This study has some limitations. First, it was a single-center and retrospective study. Second, there was no follow-up data regarding patients’ outcomes. However, the sample size was good (130), and we could obtain all clinical, biochemical, and histopathological details of these patients. This study impactfully adds to the literature on the NDKD spectrum in patients with DM.
In conclusion, patients with DM undergoing kidney biopsy have a high prevalence of NDKD (60.79%). Kidney biopsy should not be deferred in patients, as the benefits of diagnosing and treating NDKD outweigh the risks. The management and prognosis of many patients can be improved after diagnosis of NDKD by kidney biopsy.
Acknowledgements
The authors thanks Dr. Anand Chellapan for his advice in experimental design and statistical analysis.
Conflicts of interest
There are no conflicts of interest.
References
- Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care. 2004;27:1047-53.
- [CrossRef] [PubMed] [Google Scholar]
- International Diabetes Federation. (10th edition). Available from: https://diabetesatlas.org/idfawp/resource-files/2021/07/IDF_Atlas_10th_Edition_2021.pdf
- Update on diabetic nephropathy: Core curriculum 2018. Am J Kidney Dis. 2018;71:884-895.
- [CrossRef] [PubMed] [Google Scholar]
- Non diabetic renal disease in type 2 diabetes mellitus. Nephrology (Carlton). 2006;11:533-7.
- [CrossRef] [PubMed] [Google Scholar]
- Prevalence of nondiabetic renal disease in diabetic patients. Am J Nephrol. 2007;27:322-8.
- [CrossRef] [PubMed] [Google Scholar]
- Non-diabetic renal disease in patients with type-2 diabetes mellitus. Saudi J Kidney Dis Transpl. 2012;23:1000-7.
- [CrossRef] [PubMed] [Google Scholar]
- Non-diabetic renal disease (NDRD) in patients with type 2 diabetes mellitus (type 2 DM) J Assoc Physicians India. 2013;61:194-9.
- [PubMed] [Google Scholar]
- The modern spectrum of renal biopsy findings in patients with diabetes. Clin J Am Soc Nephrol. 2013;8:1718-24.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Clinical and pathological characteristics of non-diabetic renal disease in type 2 diabetes patients. Clin Kidney J. 2018;11:342-7.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Characterizing predictors of non-diabetic kidney disease (NDKD) in diabetic patients. Int Urol Nephrol. 2022;54:1303-9.
- [CrossRef] [PubMed] [Google Scholar]
- Renal biopsy in patients with diabetes: A pooled meta-analysis of 48 studies. Nephrol Dial Transplant. 2017;32:97-110.
- [CrossRef] [PubMed] [Google Scholar]
- Non-diabetic renal disease in type 2 diabetes mellitus: Study of renal - retinal relationship. Indian J Nephrol. 2015;25:222-8.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Heterogeneity in kidney histology and its clinical indicators in type 2 diabetes mellitus: A retrospective study. J Clin Med. 2023;12:1778.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Predictive factors for non-diabetic nephropathy in diabetic patients The utility of renal biopsy. Nefrologia. 2016;36:535-44.
- [CrossRef] [PubMed] [Google Scholar]
- Heterogeneous nature of renal lesions in type II diabetes. J Am Soc Nephrol. 1993;3:1458-66.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical features of non-diabetic renal diseases in patients with type 2 diabetes. Diabetes Res Clin Pract. 2005;69:237-42.
- [CrossRef] [PubMed] [Google Scholar]
- Diabetic retinopathy in predicting diabetic nephropathy in patients with type 2 diabetes and renal disease: A meta-analysis. Diabetologia. 2013;56:457-66.
- [CrossRef] [PubMed] [Google Scholar]
- Nondiabetic kidney disease in type 2 diabetic patients: A single center experience. Indian J Nephrol. 2012;22:358-62.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Histological spectrum of clinical kidney disease in type 2 diabetes mellitus patients with special reference to nonalbuminuric diabetic nephropathy: A kidney biopsy-based study. J Assoc Physicians India. 2022;70:11-2.
- [CrossRef] [PubMed] [Google Scholar]
- IgA Nephropathy complicating diabetic glomerulosclerosis. Nephron. 1998;79:279-87.
- [CrossRef] [PubMed] [Google Scholar]
- Membranous glomerulonephritis in diabetic patients: A study of 15 cases and review of the literature. Mod Pathol. 1990;3:36-42.
- [PubMed] [Google Scholar]
- Idiopathic membranous nephropathy in patients with diabetes mellitus: A diagnostic and therapeutic quandary! Clin Kidney J. 2018;11:46-50.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Prevalence of non-diabetic renal disease in type 2 diabetic patients in a diabetes centre in southern india. J Assoc Physicians India. 2002;50:1135-9.
- [PubMed] [Google Scholar]
- Non-Diabetic kidney disease in type 2 diabetes mellitus: A changing spectrum with therapeutic ascendancy. J Clin Med. 2023;12:1705.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]
- Prevalence of diabetic nephropathy complicating non-diabetic renal disease among chinese patients with type 2 diabetes mellitus. Eur J Med Res. 2013;18:4.
- [CrossRef] [PubMed] [PubMed Central] [Google Scholar]








