Translate this page into:
Oxalate Nephropathy
Address for correspondence: Dr. Praveen K. Etta, Department of Nephrology and Kidney Transplantation, Virinchi Hospitals, Hyderabad, Telangana, India. E-mail: drpraveen85@gmail.com
-
Received: ,
Accepted: ,
This article was originally published by Wolters Kluwer - Medknow and was migrated to Scientific Scholar after the change of Publisher.
Dear Editor,
Acute kidney injury (AKI) in patients with coronavirus disease 2019 (COVID-19) has an incidence of 30%–40% among hospitalized patients and is a predictor of poor prognosis and mortality.[1] We describe a case of secondary oxalate nephropathy probably caused by excessive vitamin C administration.
A 60-year-old man was hospitalized with COVID-19 pneumonia. He was hypertensive and diabetic for about 5 years. He had fever, cough, and dyspnea of 1-week duration. His baseline renal function was normal. Computed tomogram (CT) chest showed bilateral interstitial pneumonia with a moderate CT severity score (18/25). After admission to the hospital, he deteriorated rapidly, was intubated, and kept on mechanical ventilation after transferring to the intensive care unit (ICU). He was started on parenteral antibiotics, remdesivir, corticosteroids, and anticoagulants. He developed cytokine storm. He was planned for tocilizumab therapy but it was unavailable. He was started on high-dose parenteral vitamin C therapy at 50 mg/kg dose, every 8th hourly, and continued for 10 days. The clinical course was further complicated by septic shock with the need for inotropes; he developed multiorgan failure with anuric AKI in the next few days. Due to metabolic acidosis, he was initiated on slow low-efficiency dialysis. After about 10 days of ICU stay, he showed gradual recovery and was extubated. He remained oliguric thereafter, requiring intermittent hemodialysis for about another week. Repeat testing for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was negative after 2 weeks. He recovered partially from AKI and became dialysis independent. Serum creatinine remained around 4 mg/dl. Urine examination showed 1+ albuminuria, with no active sediments. Kidney biopsy was performed due to non-recovery from AKI at the end of 4 weeks. The viable glomeruli were normal. Prominent tubular injury was seen with irregular dilatation, denudation of lining epithelium, flattening, and focal cytoplasmic vacuolation. It also showed deposition of birefringent calcium oxalate crystals within few tubular lumina, best appreciated on polarized light. The interstitium was widened, fibrosed, and mildly edematous with moderate infiltrate of lymphocytes. Occasional focus showed multinucleated giant cells with calcium oxalate crystals within [figure 1]. There was mild to moderate tubular atrophy. Arteries showed mild eccentric intimal fibrosis and focal intimal myxoid change. Immunofluorescence was inconclusive. Energy-dispersive X-ray spectrometry analysis of biopsy tissue was not done in our case.
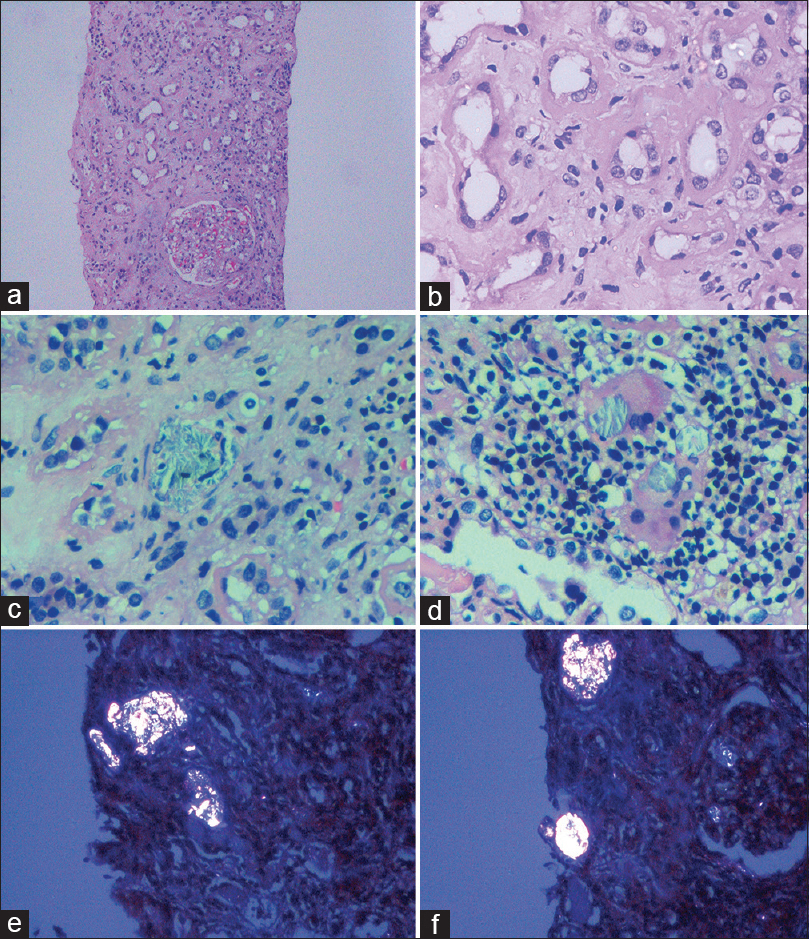
- Light microscopy examination of the kidney biopsy specimen. Low-power and high-power views showing evidence of acute tubular injury (a, ×10 and b, ×40), with calcium oxalate crystals in the lumina of some tubules (c, ×40). Crystals are also seen within multinucleated giant cells, which are surrounded by numerous lymphocytes (d, ×40). Sections examined under polarized light showing several birefringent calcium oxalate crystals in the tubular lumina (e and f, ×20) (hematoxylin and eosin stain)
The factors that contribute to AKI in COVID-19 include sepsis with altered renal microcirculation, cytokine storm, mechanical ventilation, hypoxemia, hypotension, metabolic acidosis, and drugs. The SARS-CoV-2 virus can cause direct pathological injury to kidneys, leading to acute tubular injury, acute tubular necrosis, acute interstitial nephritis, thrombotic microangiopathy, collapsing glomerulopathy, and other glomerular pathologies.[2] In our patient, though unlikely, we cannot exclude direct viral-mediated AKI as biopsy was done relatively late when he had recovered from COVID-19 pneumonia. Initial AKI could be multifactorial, which further precipitated the deposition of calcium oxalate crystals due to secondary hyperoxaluria caused by excessive vitamin C therapy. In the background of preexisting kidney dysfunction or tubular injury, intratubular retention of calcium oxalate crystals can occur and precipitate oxalate nephropathy, which in turn can worsen AKI and affect its recovery.
In a recent systematic review on secondary oxalate nephropathy, renal replacement therapy was required in >50% of patients. None had a complete recovery from AKI, 42% had partial recovery, and most patients (58%) remained dialysis-dependent; 33% of patients died. Fat malabsorption is probably the most commonly attributed cause of secondary oxalate nephropathy.[3] Vitamin C causes secondary hyperoxaluria through the endogenous conversion of ascorbic acid to oxalate.[4] It has antioxidant properties and can ameliorate the pathophysiological effects of cytokine storm and sepsis.[5] The intake of vitamin C supplements as low as 1 or 2 g per day may produce a significant increase in urinary oxalate, elevating the risk of calcium oxalate crystallization.[6] We used almost 90 g total vitamin C dose in our patient. Altered intestinal permeability to dietary oxalate may also play a role. Few antibiotics can precipitate and form crystals in the renal tubules. These may also induce precipitation of oxalate crystals in the presence of predisposing factors. Though we use vitamin C widely, this phenomenon is not widely described in the literature. Many of these cases of AKI rarely undergo kidney biopsy; thus, it could be underreported.[7] Whenever feasible, kidney biopsy should be performed in all cases of AKI with no recovery within few weeks for establishing the diagnosis and prognostication. Excessive and injudicious use of vitamin C can lead to secondary hyperoxaluria and precipitate or worsen AKI.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Acute kidney injury in patients hospitalized with COVID-19. Kidney Int. 2020;98:209-18.
- [Google Scholar]
- Acute kidney injury due to collapsing glomerulopathy following COVID-19 infection. Kidney Int Rep. 2020;5:940-5.
- [Google Scholar]
- Secondary oxalate nephropathy: A systematic review. Kidney Int Rep. 2018;3:1363-72.
- [Google Scholar]
- Effect of vitamin C infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: The CITRIS-ALI randomized clinical trial. JAMA. 2019;322:1261-70.
- [Google Scholar]
- Effect of vitamin C supplements on urinary oxalate and pH in calcium stone-forming patients. Kidney Int. 2003;63:1066-71.
- [Google Scholar]
- Oxalate nephropathy caused by excessive vitamin C administration in 2 patients with COVID-19. Kidney Int Rep. 2020;5:1815-22.
- [Google Scholar]






